Advertisements
Advertisements
प्रश्न
We would like to make a vessel whose volume does not change with temperature (take a hint from the problem above). We can use brass and iron `(β_(vbrass) = (6 xx 10^(–5))/K and β_(viron) = (3.55 xx 10^(–5))/K)` to create a volume of 100 cc. How do you think you can achieve this.
Advertisements
उत्तर
In the previous problem, the difference in the length was constant.
In this problem the difference in volume is constant.
The situation is shown in the diagram.
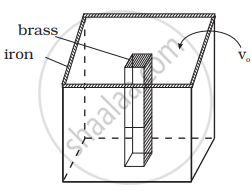
Let Vio, Vbo be the volume of iron and brass vessel at 0°C
Vi, Vb be the volume of iron and brass vessel at Δθ°C,
γi, γb be the coefficient of volume expansion of iron and brass.
As per the question, Vio – Vbo = 100 cc = Vi – Vb ......(i)
Now, `V_i = V_(io) (1 + γ_iΔθ)`
`V_b = V_(bo) (1 + γ_bΔθ)`
`V_i - V_b = (V_(io) - V_(bo)) + Δθ(V_(io)γ_i - V_(bo)γ_b)`
Since, `V_i - V_b` = constant
So, `V_(io)γ_i - V_(bo)γ_b`
⇒ `V_(io)/V_(bo) = γ_b/γ_i`
= `(3/2 β_b)/(3/2 β_i)`
= `β_b/β_i`
= `(6 xx 10^-5)/(3.55 xx 10^-5)`
= `6/3.55`
`V_(io)/V_(bo) = 6/3.55` ......(ii)
Solving equations (i) and (ii), we get
Vio = 244.9 cc
Vbo = 144.9 cc
APPEARS IN
संबंधित प्रश्न
Given below are observations on molar specific heats at room temperature of some common gases.
| Gas |
Molar specific heat (Cv) (cal mol–1 K–1) |
| Hydrogen | 4.87 |
| Nitrogen | 4.97 |
| Oxygen | 5.02 |
| Nitric oxide | 4.99 |
| Carbon monoxide | 5.01 |
| Chlorine | 6.17 |
The measured molar specific heats of these gases are markedly different from those for monatomic gases. Typically, molar specific heat of a monatomic gas is 2.92 cal/mol K. Explain this difference. What can you infer from the somewhat larger (than the rest) value for chlorine?
A child running a temperature of 101°F is given an antipyrin (i.e. a medicine that lowers fever) which causes an increase in the rate of evaporation of sweat from his body. If the fever is brought down to 98 °F in 20 min, what is the average rate of extra evaporation caused, by the drug? Assume the evaporation mechanism to be the only way by which heat is lost. The mass of the child is 30 kg. The specific heat of human body is approximately the same as that of water, and latent heat of evaporation of water at that temperature is about 580 cal g–1.
Calculate the mass of ice needed to cool 150 g of water contained in a calorimeter of mass 50 g at 32 °C such that the final temperature is 5 °C. Specific heat capacity of calorimeter = 0.4 J g-1 °C-1, Specific heat capacity of water = 4.2 J g-1°C-1, latent heat capacity of ice = 330 J g-1.
What do you understand by the following statements:
The heat capacity of the body is 60JK-1.
Heat supplied to a solid change it into liquid. What is this change in the phase called?
Differentiate between heat capacity and specific heat capacity.
Discuss the role of high specific heat capacity of water with reference to climate in coastal areas.
Find the time taken by a 500 W heater to raise the temperature of 50 kg of material of specific heat capacity 960 J kg-1K-1, from 18°C to 38° C. Assume that all the heat energy supplied by the heater is given to the material.
State the impact of global warming on life on the earth.
What is carbon tax?
How will you prove experimentally that different substances have different specific heat capacities?
In Regnault's apparatus for measuring specific heat capacity of a solid, there is an inlet and an outlet in the steam chamber. The inlet is near the top and the outlet is near the bottom. Why is it better than the opposite choice where the inlet is near the bottom and the outlet is near the top?
What is the specific heat capacity of boiling water?
Indian style of cooling drinking water is to keep it in a pitcher having porous walls. Water comes to the outer surface very slowly and evaporates. Most of energy needed for evaporation is taken from the water itself and the water is cooled down. Assume that a pitcher contains 10 kg of water and 0.2 g of water comes out per second. Assuming no backward heat transfer from the atmosphere to the water, calculate the time in which the temperature decrease by 5°C. Specific heat capacity of water = 4200 J kg−1 °C−1 and latent heat of vaporization of water = 2.27 × 106 J kg−1.
Define heat capacity.
A solid of mass 80 g at 80°C is dropped in 400 g water at 10°C. If final temp. is 30°C, find the sp. heat cap. of the solid.
Fill in the following blank using suitable word:
SI unit of heat is .........
What are other units of heat? Name and define them.
Explain, Why is it advisabile to pour cold water over burns, caused on human body, by hot solids?
Some heat is provided to a body to raise its temperature by 25°C. What will be the corresponding rise in temperature of the body as shown on the Kelvin scale?
1 kg of water freezes to form ice at 0°C. What amount of heat is withdrawn?
Why are athletes advised to put on extra clothes after competing on event?
A substance is in the form of a solid at 0°C. The amount of heat added to this substance and the temperature of the substance are plotted on the following graph:

If the specific heat capacity of the solid substance is 500 J/kg °G, find from the graph, the mass of the substance.
The temperature of a lead piece of mass 400 g rises from 20°C to 50°C when 1560 J of heat is supplied to it. Calculate Specific heat capacity of lead.
Water falls from a height of 50 m. Calculate the rise in the temperature of water when it strikes the bottom.
(g = 10 ms-2; Specific heat capacity of water = 4200 J / kg°C)
An electric immersion heater is rated 1250 W. Calculate the time in which it will heat 20 kg of water at 5°C to 65°C.
The SI unit of specific heat is _______.
All metals have the same specific heat capacity.
The heat capacity of the vessel of mass 100 kg is 8000 J/°K. Find its specific heat capacity.
Numerical Problem.
What is the heat in joules required to raise the temperature of 25 grams of water from 0°C to 100°C? What is the heat in Calories? (Specific heat of water = `(4.18"J")/("g"°"C")`
The molar specific heats of an ideal gas at constant pressure and constant volume are denoted by Cp and Cv respectively. If `gamma = "C"_"p"/"C"_"v"` and R is the universal gas constant, then Cp is equal to ______.
Two metals A and B have specific heat capacities in the ratio 2:3. If they are supplied same amount of heat then
If specific heat capacity of metal A is 0.26 Jg-1 0C-1 then calculate the specific heat capacity of metal B.
What is the amount of heat required to raise the temperature of 5 kg of iron from 30°C to 130°C? Specific heat capacity of iron = 483 Jkg-1C-1.
The molar specific heats of an ideal gas at constant pressure and volume are denoted by Cp and Cv, respectively. If `gamma = "C"_"p"/"C"_"v"` and R is the universal gas constant, then Cv is equal to ______.
Thermal capacities of substances A and B are same. If mass of A is more than mass of B then:
Which substance will have more specific heat capacity?
A mixture of one mole of monoatomic gas and one mole of a diatomic gas (rigid) are kept at room temperature (27°C). The ratio of specific heat of gases at constant volume respectively is:
