Advertisements
Advertisements
प्रश्न
We would like to make a vessel whose volume does not change with temperature (take a hint from the problem above). We can use brass and iron `(β_(vbrass) = (6 xx 10^(–5))/K and β_(viron) = (3.55 xx 10^(–5))/K)` to create a volume of 100 cc. How do you think you can achieve this.
Advertisements
उत्तर
In the previous problem, the difference in the length was constant.
In this problem the difference in volume is constant.
The situation is shown in the diagram.
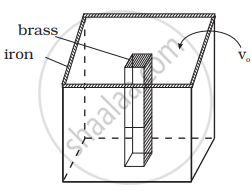
Let Vio, Vbo be the volume of iron and brass vessel at 0°C
Vi, Vb be the volume of iron and brass vessel at Δθ°C,
γi, γb be the coefficient of volume expansion of iron and brass.
As per the question, Vio – Vbo = 100 cc = Vi – Vb ......(i)
Now, `V_i = V_(io) (1 + γ_iΔθ)`
`V_b = V_(bo) (1 + γ_bΔθ)`
`V_i - V_b = (V_(io) - V_(bo)) + Δθ(V_(io)γ_i - V_(bo)γ_b)`
Since, `V_i - V_b` = constant
So, `V_(io)γ_i - V_(bo)γ_b`
⇒ `V_(io)/V_(bo) = γ_b/γ_i`
= `(3/2 β_b)/(3/2 β_i)`
= `β_b/β_i`
= `(6 xx 10^-5)/(3.55 xx 10^-5)`
= `6/3.55`
`V_(io)/V_(bo) = 6/3.55` ......(ii)
Solving equations (i) and (ii), we get
Vio = 244.9 cc
Vbo = 144.9 cc
APPEARS IN
संबंधित प्रश्न
What amount of heat must be supplied to 2.0 x 10-2 kg of nitrogen (at room temperature) to raise its temperature by 45 °C at constant pressure? (Molecular mass of N2 = 28; R = 8.3 J mol-1 K-1.)
Calculate the mass of ice needed to cool 150 g of water contained in a calorimeter of mass 50 g at 32 °C such that the final temperature is 5 °C. Specific heat capacity of calorimeter = 0.4 J g-1 °C-1, Specific heat capacity of water = 4.2 J g-1°C-1, latent heat capacity of ice = 330 J g-1.
What do you understand by the following statements:
The heat capacity of the body is 60JK-1.
State two factors upon which the heat absorbed by a body depends
During the phase change does the average kinetic energy of the molecules of the substance increase?
Give a mathematical relation between Heat Capacity and Specific Heat Capacity.
Name the S.I. unit of heat.
Give one example where high specific heat capacity of water is used as cooling purposes?
Name three fossil fuels that emit carbon dioxide into the atmosphere ?
Give three reasons for the increase of green house gases.
What is meant by global warming?
What impact will global warming have on the health of the affected population?
Who shall pay carbon tax ?
Solve the following problems:
Equal heat is given to two objects A and B of mass 1 g. Temperature of A increases by 3°C and B by 5°C. Which object has more specific heat? And by what factor?
State, with reason, which of the two, boiling water or steam both at 100°C will produce more severe burns.
If 10125 J of heat energy boils off 4.5 g of water at 100°C to steam at 100°C, find the specific latent heat of steam.
A liquid X has specific heat capacity higher than the liquid Y. Which liquid is useful as coolant in car radiators.
Water boils at 120 °C in a pressure cooker. Explain the reason.
What is heat? What is the S. I. unit of heat?
Does the specific heat capacity of a substance depend upon its mass and rise in temperature only?
Explain, why does a wise farmer water his fields, if forecast is forst?
Ice-cream at 0°C feels colder than water at 0°C. Give reason for this observation.
Will the value of specific heat’capacity and specific latent heat of a substance change if the scale is °F instead of °C?
The molar specific heat of a gas at constant volume is 12307.69 J kg-1 K-1. If the ratio of the two specific heats is 1.65, calculate the difference between the two molar specific heats of gas.
State factors on which the amount of heat radiated by a body depends.
How much heat energy is necessary to raise the temperature of 5 kg of water from 20°C to 100°C?
Read the passage and answer the questions based on it.
If heat is exchanged between a hot and cold object, the temperature of the cold object goes on increasing due to gain of energy and the temperature of the hot object goes on decreasing due to loss of energy. The change in temperature continues till the temperatures of both objects attain the same value. In this process, the cold object gains heat energy and the hot object loses heat energy. If the system of both the objects is isolated from the environment by keeping it inside a heat-resistant box then no energy can flow from inside the box or come into the box. In this situation, we get the following principle.
Heat energy lost by the hot object = Heat energy gained by the cold object. This is called the ‘Principle of heat exchange’.
- Where does heat transfer take place?
- In such a situation which principle of heat do you perceive?
- How can this principle be explained in short?
- Which property of the substance is measured using this principle?
When a uniform rod is heated, which of the following quantity of the rod will increase
For a gas, `"R"/"C"_"v"=0.4`, where R Is universal gas constant and Cv is the molar specific heat at constant volume. The gas is made up of molecules, which are ______
A diatomic gas undergoes adiabatic change. Its pressure 'P' and temperature 'T' are related as p ∝ Tx, where x is ______.
For a gas `"R"/"C"_"v" = 0.4,` where 'R' is the universal gas constant and 'Cv' is molar specific heat at constant volume. The gas is made up of molecules which are ______.
Two metals A and B have specific heat capacities in the ratio 2:3. If they are supplied same amount of heat then
If specific heat capacity of metal A is 0.26 Jg-1 0C-1 then calculate the specific heat capacity of metal B.
Why is water used as a coolant in radiators of a car?
The difference between the two molar specific heats of gas is 9000 J/kg K. If the ratio of the two specific heats is 1.5, calculate the two molar specific heats.
A geyser heats water flowing at a rate of 2.0 kg per minute from 30°C to 70°C. If the geyser operates on a gas burner, the rate of combustion of fuel will be ______ g min-1.
[Heat of combustion = 8 × 103 Jg-1 Specific heat of water = 4.2 Jg-1°C-1]
When two kilocalories of heat are supplied to a system, the internal energy of the system increases by 5030 J and the work done by the gas against the external pressure is 3350 J. Calculate J, the mechanical equivalent of heat.
A mixture of one mole of monoatomic gas and one mole of a diatomic gas (rigid) are kept at room temperature (27°C). The ratio of specific heat of gases at constant volume respectively is:
