Advertisements
Advertisements
Question
We would like to make a vessel whose volume does not change with temperature (take a hint from the problem above). We can use brass and iron `(β_(vbrass) = (6 xx 10^(–5))/K and β_(viron) = (3.55 xx 10^(–5))/K)` to create a volume of 100 cc. How do you think you can achieve this.
Advertisements
Solution
In the previous problem, the difference in the length was constant.
In this problem the difference in volume is constant.
The situation is shown in the diagram.
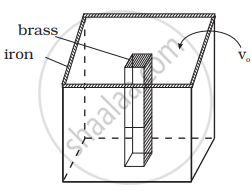
Let Vio, Vbo be the volume of iron and brass vessel at 0°C
Vi, Vb be the volume of iron and brass vessel at Δθ°C,
γi, γb be the coefficient of volume expansion of iron and brass.
As per the question, Vio – Vbo = 100 cc = Vi – Vb ......(i)
Now, `V_i = V_(io) (1 + γ_iΔθ)`
`V_b = V_(bo) (1 + γ_bΔθ)`
`V_i - V_b = (V_(io) - V_(bo)) + Δθ(V_(io)γ_i - V_(bo)γ_b)`
Since, `V_i - V_b` = constant
So, `V_(io)γ_i - V_(bo)γ_b`
⇒ `V_(io)/V_(bo) = γ_b/γ_i`
= `(3/2 β_b)/(3/2 β_i)`
= `β_b/β_i`
= `(6 xx 10^-5)/(3.55 xx 10^-5)`
= `6/3.55`
`V_(io)/V_(bo) = 6/3.55` ......(ii)
Solving equations (i) and (ii), we get
Vio = 244.9 cc
Vbo = 144.9 cc
APPEARS IN
RELATED QUESTIONS
A solid of mass 50 g at 150 °C is placed in 100 g of water at 11 °C when the final temperature recorded is 20 °C. Find the specific heat capacity of the solid. (specific heat capacity of water = 4.2 J/g °C)
Calculate the mass of ice needed to cool 150 g of water contained in a calorimeter of mass 50 g at 32 °C such that the final temperature is 5 °C. Specific heat capacity of calorimeter = 0.4 J g-1 °C-1, Specific heat capacity of water = 4.2 J g-1°C-1, latent heat capacity of ice = 330 J g-1.
Differentiate between heat capacity and specific heat capacity.
Name a liquid which has the highest specific heat capacity.
What do you mean by the following statement?
The specific heat capacity of copper is 0. 4 Jg-1 K-1?
Water is used in hot water bottles for fomentation. Give a reason.
A mass m1 of a substance of specific heat capacity c1 at temperature t1 is mixed with a mass m2 of other substance of specific heat capacity c2 at a lower temperature t2. Deduce the expression for the temperature t of the mixture. State the assumption made, if any.
How much heat energy is released when 5.0 g of water at 20℃ changes into ice at 0℃? Take specific heat capacity of water = 4.2 J g-1 K-1, Specific latent heat of fusion of ice = 336 J g-1.
Name the radiations for which the green house gases are transparent ?
What is meant by global warming?
Who shall pay carbon tax ?
A calorimeter has mass 100 g and specific heat 0.1 kcal/ kg °C. It contains 250 gm of liquid at 30°C having specific heat of 0.4 kcal/kg °C. If we drop a piece of ice of mass 10 g at 0°C, What will be the temperature of the mixture?
Figure shows a paddle wheel coupled to a mass of 12 kg through fixed frictionless pulleys. The paddle is immersed in a liquid of heat capacity 4200 J K−1 kept in an adiabatic container. Consider a time interval in which the 12 kg block falls slowly through 70 cm. (a) How much heat is given to the liquid? (b) How much work is done on the liquid? (c) Calculate the rise in the temperature of the liquid neglecting the heat capacity of the container and the paddle.

Give one example where high specific heat capacity of water is used as cooling.
Define heat capacity.
State the condition for the flow of heat energy from one body to another.
m kg of a substance of specific heat capacity s J/kg °C is heated so that its temperature rises from θ1°C to θ2°C. Write down the expression for the heat Q supplied.
Why are athletes advised to put on extra clothes after competing on event?
Describe a method to determine the specific heat capacity of a solid (say, a piece of copper).
Discuss how high specific heat capacity of water helps in formation of land and sea breeze.
Water falls from a height of 50 m. Calculate the rise in the temperature of water when it strikes the bottom.
(g = 10 ms-2; Specific heat capacity of water = 4200 J / kg°C)
Derive Mayer’s relation.
For a gas, `"R"/"C"_"v"=0.4`, where R Is universal gas constant and Cv is the molar specific heat at constant volume. The gas is made up of molecules, which are ______
50 g of copper is heated to increase its temperature by 10° C. If the same quantity of heat is given to 5 g water, the rise in its temperature is [Specific heat of copper = 420 joule-kg-1 °C-1 , specific heat of water = 4200 joule-kg-I °C-1]
Which of the following substances (A, B and C) has the highest specific beat?

The diagram below shows a cooling curve for 200 g of water. The heat is extracted at the rate of 100 Js-1. Answer the questions that follow:

- Calculate specific heat capacity of water.
- Heat released in the region BC.
Match the following:
| Column A | Column B | ||
| 1. | Specific heat capacity of water | a. | 0°C |
| 2. | Latent heat of fusion of ice | b. | 2260 J/g |
| 3. | Latent heat of vaporization of water | c. | 100°C |
| 4. | The melting point of iced | d. | 4.2 J/g°C |
| 5. | The boiling point of water | e. | 336 J/g |
Which of the following substances (A, B and C) has the highest specific heat?

Find the odd one out:
A geyser heats water flowing at a rate of 2.0 kg per minute from 30°C to 70°C. If the geyser operates on a gas burner, the rate of combustion of fuel will be ______ g min-1.
[Heat of combustion = 8 × 103 Jg-1 Specific heat of water = 4.2 Jg-1°C-1]
Observe the following diagram and answer the questions given below:

Specific heat capacity of metals
- Which element has maximum specific heat capacity? Justify.
- Which element has minimum specific heat capacity? Justify.
- Define specific heat of object.
Give one example where high specific heat capacity of water is used as heat reservoir.
Which method is used to determine the specific heat capacity of both solids and liquids by mixing a substance of unknown specific heat with water?
