Topics
Chemical Reactions and Equations
- Chemical Reactions in Daily Life
- Chemical Equations
- Balancing Chemical Equation
- Types of Chemical Reactions > Combination Reaction
- Types of Chemical Reactions > Decomposition Reaction
- Types of Chemical Reactions > Single Displacement Reaction
- Types of Chemical Reactions > Double Displacement Reaction
- Chemical Properties of Carbon Compounds > Oxidation
- The Effects of Oxidation Reactions in Everyday Life
Acids, Bases and Salts
- Acids and Bases in Daily Life
- Acids and Bases in the Laboratory
- Acids and Bases React with Metals
- Reaction of Metal Carbonates with Acids
- Acids and Bases Reaction with each other
- Reaction of Metallic Oxides with Acids
- Reaction of a Non-metallic Oxide with Base
- Common Properties of Acids and Bases
- The pH Scale
- Importance of pH in Everyday Life
- Salts > Family of Salts
- Salts > pH of Salts
- Salts > Chemicals from Common Salt
- Salts > Water in Salt Crystals
Metals and Non-metals
Carbon and its Compounds
- Importance of Carbon
- The Covalent Bond
- Allotropes of Carbon > Diamond
- Allotropes of Carbon > Graphite
- Allotropes of Carbon > Fullerene
- Carbon: A Versatile Element
- Organic Compounds
- Classification of Hydrocarbons
- Carbon Compounds: Chains, Branches, Rings
- Homologous Series
- Nomenclature
- Chemical Properties of Carbon Compounds > Combustion
- Ethanol
- Ethanoic Acid
- Soaps and Detergents
Life Processes
- Life Processes in Living Organisms
- Nutrition
- Autotrophic Nutrition
- Heterotrophic Nutrition
- Nutrition in Human Beings
- Dental Caries
- Production of ATP
- Blood Circulatory System
- Blood Vessels Entering and Leaving The Heart
- Valves of the Heart
- Tissue Fluid (Or Intercellular Fluid)
- Transportation in Plants
- Transportation of Water
- Excretion
- Excretion in Human Beings
- Kidney and Its Internal Structure
- Structure of a Kidney Tubule (Nephrons)
- Dialysis and Artificial Kidney
- Excretion in Plants
- Organ and Body Donation
Control and Co-ordination
How do Organisms Reproduce?
Heredity
Light – Reflection and Refraction
- Light and Its Straight-Line Propagation
- Reflection of Light
- Spherical Mirrors
- Image Formation by Spherical Mirrors
- Representation of Images Formed by Spherical Mirrors
- Image Formation by Concave Mirror
- Image Formation by a Convex Mirror
- Sign Convention for Reflection by Spherical Mirrors
- Ray Optics - Mirror Formula
- Refraction of Light
- Refraction through a Rectangular Glass Slab
- The Refractive Index
- Refraction by Spherical Lenses
- Image Formation by Lenses
- Image Formation in Lenses Using Ray Diagrams
- Sign Convention for Spherical Lenses
- Lens Formula
- Power of a Lens
The Human Eye and the Colourful World
- Human Eye
- Defects of Vision and Their Correction
- Defects of Vision and Their Corrections > Myopia
- Defects of Vision and Their Corrections > Hypermetropia
- Defects of Vision and Their Corrections > Presbyopia
- Refraction of Light Through a Prism
- Dispersion of Light
- Atmosphere Refraction
- Scattering of Light
Electricity
Magnetic Effects of Electric Current
- Magnetic Effect of Electric Current
- Applications of Biot-Savart's Law > Magnetic Field due to a Finite Straight Current-Carrying Wire
- Magnetic Field Due to a Current-Carrying Conductor
- Right-hand Thumb Rule
- Applications of Biot-Savart's Law > Magnetic Field at the Centre of a Circular Loop
- Applications of Ampere’s Circuital Law > Magnetic Field of a Long Straight Solenoid
- Force on a Current Carrying Conductor in a Magnetic Field
- Fleming’s Left Hand Rule
- Magnetism in Medicine
- Domestic Electric Circuits
Our Environment
- Definition: Chemical Equation
- Key Points: Chemical Equations
CISCE: Class 10
Definition: Chemical Equation
A chemical equation is a balanced account of a chemical transaction. It is not merely a qualitative statement, but it also gives quantitative information of a chemical reaction.
OR
The representation of a chemical reaction in a condensed form using chemical formulae is called as the chemical equation.
Maharashtra State Board: Class 10
Key Points: Chemical Equations
- Word equations use names; chemical equations use formulas.
- Reactants → Products, with arrow showing reaction direction.
- Use + between two or more reactants or products.
- Show states: (s), (l), (g), (aq); use ↑ for gas, ↓ for precipitate.
- Heat (Δ) or other conditions go above/below the arrow.
Introduction
A chemical equation represents a chemical reaction in a concise form using chemical symbols and formulas. It shows the transformation of reactants into products with proper notations.
Word Equation and Chemical Equation:
- A chemical reaction can first be written in words, called a word equation.
Example:
Copper sulfate solution + Zinc → Zinc sulfate solution + Copper - A more condensed representation using chemical formulas is called a chemical equation.
Example: CuSO₄ + Zn → ZnSO₄ + Cu
Writing a Chemical Equation
Reactants are written on the left and products on the right, separated by an arrow (→) indicating the direction of the reaction.
Multiple reactants or products are separated by a plus sign (+).
Physical states are indicated using symbols:
- Solid: (s), Liquid: (l), Gas: (g), Aqueous solution: (aq).
- A gas product can also be indicated with an upward arrow (↑), and an insoluble solid (precipitate) with a downward arrow (↓).
Example with states: CuSO₄(aq) + Zn(s) → ZnSO₄(aq) + Cu(s)
Heat and Reaction Conditions:
- When heat is required for a reaction, the symbol Δ (delta) is placed above the arrow.
Example:
CaCO₃(s) → ΔCaO(s) + CO₂↑ - If heat is released during a reaction, it is explicitly written.
Example:
CuSO₄(aq) + Zn(s) → ZnSO₄(aq) + Cu(s) + Heat - Special conditions such as temperature, pressure, or catalysts are mentioned above or below the arrow.
Example:
\[\mathrm{Vegetable~oil~(l)+H_2(g)}\quad\frac{60^oC}{\text{Ni Catalyst}}\quad\mathrm{Vanaspathi~ghee~(s)}\]
Examples of Reactions:
- Reaction with concentrated nitric acid
Cu(s) + 4HNO₃(aq) → Cu(NO₃)₂(aq) + 2NO₂(g) + 2H₂O(l)
(Nitrogen dioxide (NO₂) is a reddish poisonous gas.) - Reaction with dilute nitric acid
3Cu(s) + 8HNO₃(aq) → 3Cu(NO₃)₂(aq) + 2NO(g) + 4H₂O(l)
(Nitric oxide (NO) gas is formed instead of NO₂)
Experiment
1. Aim: To observe the formation of a precipitate in a chemical reaction and verify the law of conservation of mass.
2. Requirements
- Apparatus: test tube, conical flask, balance, thread, rubber cork.
- Chemicals: Sodium chloride solution (NaCl) and silver nitrate solution (AgNO₃).
3. Procedure
- Take a conical flask and fill it with sodium chloride solution.
- Fill a test tube with silver nitrate solution and tie a thread to it.
- Carefully insert the test tube into the conical flask without mixing the solutions and seal the flask with a rubber cork to make it airtight.
- Weigh the conical flask using a balance and record the mass.
- Tilt the conical flask to mix the silver nitrate solution with the sodium chloride solution.
- Observe any visible changes, such as the formation of a precipitate.
- Weigh the conical flask again and compare it with the initial mass.
\[\begin{array}
{c}\mathrm{AgNO}_3(\mathrm{aq})+\mathrm{NaCl}(\mathrm{aq})\longrightarrow\mathrm{AgCl}\downarrow+\mathrm{NaNO}_3(\mathrm{aq}) \\
\end{array}\]
(white)
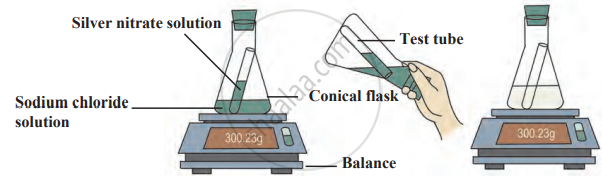
The reaction of sodium chloride with silver nitrate
4. Observation
- A white precipitate of silver chloride (AgCl) forms, indicating a chemical reaction.
- The total mass remains unchanged before and after the reaction.
5. Conclusion: The reaction between silver nitrate and sodium chloride results in the formation of an insoluble white precipitate of silver chloride. However, the total mass remains constant, confirming the law of conservation of mass.
Video Tutorials
Shaalaa.com | Chemical Reactions & Equations Part 1
Related QuestionsVIEW ALL [427]
Match the columns.
| Reactants | Products | Types of chemical reaction | ||
| 1. | MgH2 | → | Mg + H2 | Endothermic |
| 2. | 2H2S + SO2 | → | 3S + 2H2O | Oxidation |
| 3. | CaO + H2O | → | Ca(OH)2 + heat | Exothermic |
| Redox | ||||
 Na2SO4 + H2O
Na2SO4 + H2O