Topics
Some Basic Concepts of Chemistry
- Chemistry
- Development of Chemistry
- Importance and Scope of Chemistry
- States of Matter
- Classification of Matter
- Need for Measurement
- Properties of Matter and Their Measurement
- The International System of Units (SI)
- Unit and Its Types
- Unit Prefixes
- Uncertainty in Measurement
- Scientific Notation
- Significant Figures
- Accuracy, Precision and Uncertainty in Measurement
- Dimensional Analysis
- Laws of Chemical Combination
- Law of Conservation of Mass
- Law of Constant Proportions (Law of Definite Proportions)
- Law of Multiple Proportions
- Law of Reciprocal Proportions
- Gay-Lussac's Law
- Avogadro's Law
- Dalton's Atomic Theory
- Atomic Mass
- Molecular Mass
- Formula Mass
- Mole Concept and Molar Mass
- Percentage Composition, Empirical and Molecular Formula
- Stoichiometry and Stoichiometric Calculations
- Limiting Reagent
- Reactions in Solutions
Class 11
Class 12
Atomic Structure
- History of Atom
- Electrons
- Protons
- Neutrons
- Atomic Models
- J. J. Thomson’s Atomic Model
- Lord Rutherford’s Atomic model
- Drawbacks of Rutherford Atomic Model
- Atomic Number (Z) and Mass Number (A)
- Isobars
- Isotopes
- Developments Leading to the Bohr’s Atomic Model
- Wave Nature of Electromagnetic Radiation
- Particle Nature of Electromagnetic Radiation: Planck's Quantum Theory of Radiation
- The Photoelectric Effect
- Evidence for the Quantized Electronic Energy Levels - Atomic Spectra
- Bohr’s Model for Hydrogen Atom
- Hydrogen Spectrum
- Limitations of Bohr's Model
- Towards Quantum Mechanical Model of the Atom
- Dual Behaviour of Matter: De Broglie's relationship
- Heisenberg’s Uncertainty Principle
- Quantum Mechanical Model of Atom
- Quantum Mechanical Model of the Atom - Orbitals and Quantum Numbers
- Quantum Mechanical Model of the Atom - Shapes of Atomic Orbitals
- Quantum Mechanical Model of the Atom - Energies of Orbitals
- Quantum Mechanical Model of the Atom - Filling of Orbitals in Atom
- Quantum Mechanical Model of the Atom - Electronic Configuration of Atoms
- Quantum Mechanical Model of the Atom - Stability of Completely Filled and Half Filled Subshells
- Quantum Mechanical Model of the Atom - Concept of Shells and Subshells
- Structure of the Atom and Nucleus
Chemical Bonding and Molecular Structure
- Concept of Chemical Bonding
- Periodic Trends in the Modern Periodic Table
- Kossel and Lewis Approach to Chemical Bonding
- Kossel-lewis Approach to Chemical Bonding - Octet Rule
- Kossel-lewis Approach to Chemical Bonding - Covalent Bond
- Lewis Structures (Lewis Representation of Simple Molecules)
- Formal Charge
- Limitations of the Octet Rule
- Ionic or Electrovalent Bond
- Bond Parameters
- Bond Length
- Bond Enthalpy
- Bond Angle
- Bond Order
- Resonance Structures
- Polarity of Bonds
- Dipole Moment
- Valence Shell Electron Pair Repulsion (VSEPR) Theory
- Valence Bond Theory (VBT)
- Valence Bond Theory - Orbital Overlap Concept
- Valence Bond Theory - Directional Properties of Bonds
- Valence Bond Theory - Overlapping of Atomic Orbitals
- Valence Bond Theory - Types of Overlapping and Nature of Covalent Bonds
- Valence Bond Theory - Strength of Sigma (σ) bond and pi (π) bond
- Hybridisation
- Types of Hybridisation
- Other Examples of sp3, sp2 and sp Hybridisation
- Hybridisation of Elements Involving d Orbitals
- Molecular Orbital Theory
- Formation of Molecular Orbitals - Linear Combination of Atomic Orbitals (LCAO)
- Conditions for the Combination of Atomic Orbitals
- Types of Molecular Orbitals
- Energy Level Diagram for Molecular Orbitals
- Electronic Configuration and Molecular Behaviour
- Bonding in Some Homonuclear Diatomic Molecules
- Magnetic Nature of the Compounds
- Hydrogen Bonding
- Cause of Formation of Hydrogen Bond
- Types of Hydrogen Bonding
- Effects of Hydrogen Bonding
- Metallic Bond
Chemical Thermodynamics
- Introduction of Chemical Thermodynamics
- Thermodynamic Terms
- The State of the System
- Classification of Thermodynamic Processes
- First Law of Thermodynamics
- Internal Energy and Change in Internal Energy
- Thermodynamics Applications
- Enthalpy (H)
- Calorimetric Measurement of ΔU and ΔH
- Standard Enthalpy of Reactions
- Enthalpy Changes During Phase Transformations
- Standard Enthalpy of Formation
- Thermochemical Equations
- Hess’ Law of Constant Heat Summation
- Enthalpies for Different Types of Reactions
- Spontaneity
- Is Decrease in Enthalpy a Criterion for Spontaneity
- Entropy and Spontaneity
- Gibbs Energy and Spontaneity
- Entropy and Second Law of Thermodynamics
- Absolute Entropy and Third Law of Thermodynamics
- Gibbs Energy Change and Equilibrium
Equilibrium
- Concept of Equilibrium
- Equilibrium in Physical Processes
- Solid-liquid Equilibrium
- Liquid-vapour Equilibrium
- Solid - Vapour Equilibrium
- Equilibrium Involving Dissolution of Solid in Liquids
- Equilibrium Involving Dissolution of Gases in Liquids
- General Characteristics of Equilibria Involving Physical Processes
- Equilibrium in Chemical Processes - Dynamic Equilibrium
- Law of Chemical Equilibrium and Equilibrium Constant
- Homogeneous Equilibria
- Heterogeneous Equilibria
- Applications of Equilibrium Constants
- Predicting the Extent of a Reaction
- Predicting the Direction of the Reaction
- Calculating Equilibrium Concentrations
- Relationship Between Equilibrium Constant K, Reaction Quotient Q and Gibbs Energy G
- Factors affecting equilibria
- Effect of Inert Gas Addition
- Introduction of Chemical Equilibrium
- Effect of Concentration Change
- Effect of Pressure Change
- Effect of Temperature Change
- Effect of change in volume
- Effect of Catalyst
- Le Chatelier's Principle
- Ionic Equilibrium in Solution
- Concept of Acid, Base, and Salt
- Arrhenius Concept of Acids and Bases
- The Brönsted-Lowry Concept of Acids and Bases
- Lewis Concept of Acids and Bases
- Ionization of Acids and Bases
- The Ionization Constant of Water and Its Ionic Product
- The pH Scale
- Ionization Constants of Weak Acids
- Ionization of Weak Bases
- Relation Between Ka and Kb
- Relation Between Ka and Kb
- Di- and Polybasic Acids and Di- and Polyacidic Bases
- Factors Affecting Acid Strength
- Hydrolysis of Salts and the Ph of Their Solutions
- Buffer Solutions
- Concept of Solubility Equilibria of Sparingly Soluble Salts
- Solubility Product Constant
- Common Ion Effect on Solubility of Ionic Salts
Redox Reactions and Electrochemistry
- Concept of Redox Reactions
- Classical Idea of Redox Reactions - Oxidation and Reduction Reactions
- Metallic Conductors and Electrolytic Conductors
- Conductance of Electrolytic Solutions
- Redox Reactions in Terms of Electron Transfer Reactions - Introduction
- Redox Reactions in Terms of Electron Transfer Reactions - Competitive Electron Transfer Reactions
- Oxidation Number
- Types of Redox Reactions
- Balancing of Redox Reactions
- Redox Reactions as the Basis for Titrations
- Limitations of Concept of Oxidation Number
- Balancing of Redox Reactions
- Redox Reactions and Electrode Processes
- Kohlrausch's law
- Faraday's Laws of Electrolysis
- Electrochemical Cells
- Nernst Equation
Classification of Elements and Periodicity in Properties
- Significance of Classification of Elements
- Genesis of Periodic Classification
- The Modern Periodic Table
- Insights into Mendeleev’s Periodic Table
- Modern Periodic Law and the Present Form of the Periodic Table
- Nomenclature of Elements with Atomic Number Greater than 100
- Electric Configurations of Elements and the Periodic Table
- Electronic Configurations and Types of Elements: S-, P-, D-, F- Blocks
- The s-Block Elements
- The p-Block Elements
- The d-Block Elements (Transition Elements)
- The f-Block Elements (Inner-transition Elements)
- Metals, Non-metals and Metalloids
- Periodic Trends in Properties of Elements
- Trends in Physical Properties
- Atomic Radius
- Ionic Radius
- Ionization Enthalpy or Ionization Energy (IE) or Ionization Potential (IP)
- Electron Gain Enthalpy
- Electronegativity
- Valency
- IUPAC Nomenclature of Elements
- Periodicity of Valence or Oxidation States
- Anomalous Properties of Second Period Elements
- Periodic Trends and Chemical Reactivity
- Structure of the Modern Periodic Table
- Study of Specific Groups in Periodic Table
- Physical Properties of Halogen Derivatives
- Atomic Size
Purification and Characteristics of Organic Compounds
- Purification of Organic Compounds
- Chromatography Method
- Chromatography Method > Adsorption Chromatography
- Chromatography Method > Partition Chromatography
- Qualitative Analysis of Organic Compounds
- Estimation of Carbon and Hydrogen
- Estimation of Nitrogen
- Estimation of Halogen
- Estimation of Sulphur
- Estimation of Phosphorous
- Important methods for determination of elements qualitatively and quantitatively
Some Basic Principles of Organic Chemistry
- Organic Chemistry
- Tetravalence of Carbon - Shapes of Organic Compounds
- Some Characteristic Features of π Bonds
- Structural Representation of Organic Compounds
- Complete, Condensed and Bond-line Structural Formulas
- Three-dimensional Representation of Organic Molecules
- Classification of Organic Compounds
- Nomenclature of Organic Compounds
- Homologous Series
- The IUPAC System of Nomenclature
- IUPAC Nomenclature of Alkanes
- Nomenclature of Organic Compounds having Functional Group(s)
- Nomenclature of Substituted Benzene Compounds
- Isomerism
- Fission of a Covalent Bond
- Substrate and Reagent
- Electron Movement in Organic Reactions
- Electron Displacement Effects in Covalent Bonds
- Inductive Effect
- Resonance Structure
- Resonance Effect
- Electromeric Effect (E Effect)
- Hyperconjugation
- Types of Organic Reactions and Mechanisms
- Methods of Purification of Organic Compounds
- Sublimation
- Crystallisation Method
- Fractional Distillation Method
- Steam Distillation
- Differential Extraction
- Chromatography Method
- Qualitative Analysis of Organic Compounds
- Qualitative Analysis of Organic Compounds - Detection of Other Elements
- Quantitative Analysis of Carbon and Hydrogen
- Quantitative Analysis of Nitrogen
- Quantitative Analysis of Halogens
- Quantitative Analysis of Sulphur
- Quantitative Analysis of Phosphorus
- Quantitative Analysis of Oxygen
- Bond Fission
Hydrocarbons
- Hydrocarbons
- Classification of Hydrocarbons
- Alkanes
- Nomenclature and Isomerism
- Preparation of Alkanes
- Physical Properties of Alkanes
- Chemical Properties of Alkanes
- Conformations (Ethane)
- Mechanism for the Monochlorination of Methane
- Alkenes
- Structure of Double Bond (Ethene)
- Nomenclature
- Isomerism in Alkenes
- Preparation of Alkenes
- Physical Properties of Alkenes
- Chemical Properties of Alkenes
- Stereochemistry of Electrophilic Addition
- Alkynes
- Nomenclature and Isomerism
- Structure of Triple Bond
- Preparation of Alkynes
- Physical Properties of Alkynes
- Chemical Properties of Alkynes
- Addition of Hydrogen Halides to Alkyne
- Aromatic Hydrocarbons
- Nomenclature and Isomerism
- Structure of Benzene
- Aromaticity (Huckel Rule)
- Preparation of Benzene
- Physical Properties of Aromatic Hydrocarbons
- Chemical Properties of Aromatic Hydrocarbons
- Directive Influence of a Functional Group in Monosubstituted Benzene
- Electrophilic Substitution Reactions
- Concept of Reactivity in Electrophilic Substitution ESR
- Oxidation of the Side Chain
- Carcinogenicity and Toxicity
Solutions
- Introduction to Solution
- Types of Solutions
- Expressing Concentration of Solutions
- Solubility
- Solubility of a Solid in a Liquid
- Solubility of a Gas in a Liquid
- Vapour Pressure of Liquid Solutions
- Vapour Pressure of Liquid- Liquid Solutions
- Raoult’s Law as a Special Case of Henry’s Law
- Vapour Pressure of Solutions of Solids in Liquids
- Ideal and Non-ideal Solutions
- Relative Lowering of Vapour Pressure
- Elevation of Boiling Point
- Depression of Freezing Point
- Osmosis and Osmotic Pressure
- Reverse Osmosis and Water Purification
- Isotonic, Hypertonic and Hypotonic Solutions
- Abnormal Molar Masses
- Van't Hoff Factor
Chemical Kinetics
- Concept of Chemical Kinetics
- Rate of Chemical Reaction
- Factors Influencing Rate of a Reaction
- Dependence of Rate on Reactant Concentrations: Rate Law and Rate Constant
- Order of a Reaction
- Molecularity of Reaction
- Units of Rate Constant
- Integrated Rate Equations
- Zero Order Reactions
- First Order Reactions
- Half Life Period of a Reaction
- Pseudo First Order Reaction
- Temperature Dependence of the Rate of a Reaction
- Effect of Catalyst on the Rate of Reaction
- Collision Theory of Chemical Reactions
p-Block Elements
- Group 13 Elements - The Boron Family
- Important Compounds of Boron
- Important Trends and Anomalous Properties of Boron
- Group 14 Elements - The Carbon Family
- Group 15 Elements - The Nitrogen Family
- Group 16 Elements - The Oxygen Family
- Group 17 Elements - The Halogen Family
- Group 18 Elements - The Noble gas Family
d and f-block Elements
- General Introduction of "d" and "f" Block Element
- Position in the Periodic Table - d-block Elements
- Electronic Configurations of the d-block Elements
- General Properties of the Transition Elements (d-block)
- Important Compounds of Transition Elements
- F-block Elements
- The Lanthanoids
- The Actinoids
- Applications of d and f Block Elements
Coordination Compounds
- Concept of Coordination Compounds
- Werner’s Theory of Coordination Compounds
- Important Terms Pertaining to Coordination Compounds
- Nomenclature of Coordination Compounds
- Isomerism in Coordination Compounds
- Bonding in Coordination Compounds
- Valence Bond Theory (VBT)
- Magnetic Properties of Coordination Compounds
- Crystal Field Theory (CFT)
- Colour in Coordination Compounds
- Bonding in Metal Carbonyls
- Importance and Applications of Coordination Compounds
Organic Compounds Containing Halogens
- Introduction to Haloalkanes and Haloarenes
- Classification of Haloalkanes and Haloarenes
- Nomenclature of Haloalkanes and Haloarenes
- Nature of C-X Bond
- Methods of Preparation of Haloalkanes
- Methods of Preparation of Haloarenes
- Physical Properties of Haloalkanes
- Reactions of Haloalkanes - Nucleophilic Substitution Reactions
- Reactions of Haloalkanes - Elimination Reactions
- Reactions of Haloalkanes - Reaction with Metals
- Reactions of Haloarenes - Nucleophilic Substitution
- Reactions of Haloarenes - Electrophilic Substitution Reactions
- Reactions of Haloarenes - Reaction with Metals
- Polyhalogen Compounds
- Uses & Environmental Effects of Important Compounds
Organic Compounds Containing Oxygen
- Alcohols, Phenols and Ethers
- Classification of Alcohols, Phenols and Ethers
- Structures of Functional Groups of Alcohols, Phenols and Ethers
- Preparation of Alcohols and Phenols
- Physical Properties of Alcohols and Phenols
- Chemical Reactions of Alcohols and Phenols
- Preparation of Commercially Important Alcohols
- Preparation of Ethers
- Physical Properties of Ethers
- Chemical Properties of Ethers
- Chemical Reaction of Ethers - Cleavege of C-O Bonds
- Chemical Reaction of Ethers - Electrophilic Substitution
- Concepts of Aldehydes, Ketones, and Carboxylic Acids
- Nomenclature of Aldehydes and Ketones
- Structure of Carbonyl Group
- Preparation of Aldehydes and Ketones
- Physical Properties of Aldehydes and Ketones
- Chemical Reactions of Aldehydes and Ketones - Nucleophilic Addition Reactions
- Chemical Reactions of Aldehydes and Ketones - Reduction
- Chemical Reactions of Aldehydes and Ketones - Oxidation
- Chemical Reactions of Aldehydes and Ketones - Reactions Due to α-hydrogen
- Chemical Reactions of Aldehydes and Ketones - Other Reactions
- Uses of Aldehydes and Ketones
- Nomenclature and Structure of Carboxyl Group
- Physical Properties of Carboxylic Acids
- Chemical Reactions of Carboxylic Acids - Reactions Involving Cleavege of O-H Bond
- Chemical Reactions of Carboxylic Acids - Reactions Involving Cleavege of C-OH Bond
- Chemical Reactions of Carboxylic Acids - Reactions Involving –COOH Group
- Chemical Reactions of Carboxylic Acids - Substitution Reactions in the Hydrocarbon Part
- Uses of Carboxylic Acids
Organic Compounds Containing Nitrogen
- Introduction of Organic Compounds Containing Nitrogen
- Concept of Amines
- Classification of Amines
- Structure of Amines
- Nomenclature of Animes
- Physical Properties of Amines
- Preparation of Amines
- Chemical Reactions of Amines - Basic Character of Amines
- Chemical Reactions of Amines - Alkylation and Acylation
- Chemical Reactions of Amines - Carbylamine Reaction
- Chemical Reactions of Amines - Reaction with Nitrous Acid
- Chemical Reactions of Amines - Reaction with Arylsulphonyl Chloride
- Chemical Reactions of Amines - Electrophilic Substitution
- Identification of Primary, Secondary and Tertiary Amines
- Uses of Amines
- Diazonium Salts
- Diazonium Salts
- Method of Preparation of Diazonium Salts
- Chemical Reaction of Diazonium Salts - Reactions Involving Displacement of Nitrogen
- Chemical Reaction of Diazonium Salts - Reactions Involving Retention of Diazo Group
- Importance of Diazonium Salts in Synthesis of Aromatic Compounds
Biomolecules
- Concept of Biomolecules
- Classification of Carbohydrates
- Biomolecules in the Cell > Carbohydrates
- Preparation of Glucose
- Fructose
- D- and L-configuration
- Haworth Representation
- Structures of Glucose
- Sucrose
- Lactose
- Maltose
- Importance of Carbohydrates
- Amylose
- Amylopectin
- Cellulose
- Biomolecules in the Cell > Proteins
- Amino Acids
- Peptide Bond
- Classification of Proteins on the Basis of Molecular Shape
- Structure of Proteins
- Denaturation of Proteins
- Biomolecules in the Cell > Enzymes
- Biomolecules in the Cell > Hormones
- Biomolecules in the Cell > Vitamins
- Classification of Vitamins
- Important Vitamins, Their Sources and Their Deficiency Diseases
- Biomolecules in the Cell > Nucleic Acids
- Chemical Composition of Nucleic Acids
Principles Related to Practical Chemistry
- Detection of Elements: Lassaigne's Test
- Detection of Nitrogen
- Detection of Sulfur
- Detection of Halogen
- Detection of Phosphorus
- Chemistry Involved in Preparation of Inorganic Compounds
- p-Nitro Acetanilide
- Aniline Yellow
- Titration
- Concept of Redox Reactions
- Redox Titrations
- Tests for Acidic Hydrogen and Unsaturation
- Distinguishing Test of Terminal and Non-Terminal Alkynes
- Detection of Alcohols
- Iodoform Test
- Detection of Phenols
- Tests for Aldehydes
- Tests for Acids, Esters and Amides
- Test for Amines
- Hofmann Mustard Oil Test
- Hinsberg Reagent
- Mulliken- Barker's Test: Test of Nitro group
- Test for Carbohydrates and Sugars
- Test for Proteins
- Tests for Ketones
- Tests for Aromatic Alcohol (Phenol)
- Identification of Functional Groups
- Qualitative Analysis> Systematic Analysis of Anions
- Confirmatory Test for Anions
- Analysis of Cations
- Analysis of Zero Group Cation
- Analysis of Group-I Cations
- Analysis of Group-II Cations
- Analysis of Group-II A Cations
- Analysis of Group-II B Cations
- Analysis of Group-III Cations
- Analysis of Group-IV Cations
- Analysis of Group-V Cations
- Analysis of Group-VI Cations
- Confirmatory Tests for Cations
- Enthalpy of Solution of Solid Copper Sulphate
- Enthalpy of Neutralisation of a Strong Acid with Strong Base
- Crystalloids and Colloids
- Dispersed Phase and Dispersion Medium
- Lyophilic and Lyophobic Sols
- Study of Rate of Reaction between Hydrogen peroxide and Iodine Ion
States of Matter: Gases and Liquids
- Matter (Substance)
- States of Matter
- The Solid State
- The Liquid State
- The Gaseous State
- Intermolecular Forces
- Dipole - Dipole Forces
- Dipole-induced Dipole Forces
- Dispersion Forces Or London Forces
- Hydrogen Bond
- Intermolecular Forces Vs. Thermal Interactions
- Melting and Boiling Points
- The Gaseous State
- Gas Laws
- Boyle’s Law (Pressure - Volume Relationship)
- Charles’ Law (Temperature - Volume Relationship)
- Gay Lussac’s Law (Pressure- Temperature Relationship)
- Ideal Gas Equation
- Dalton’s Law of Partial Pressures
- Kinetic Molecular Theory of Gases
- Behaviour of Real Gases: Deviation from Ideal Gas Behaviour
- Liquefaction of Gases
- Liquid State
- Vapour Pressure
- Viscosity
- Surface Tension
s-block Elements (Alkali and Alkaline Earth Metals)
- Introduction of s-block elements
- Group 1 Elements - Alkali Metals
- General Characteristics of the Compounds of the Alkali Metals
- Anomalous Properties of Lithium
- Some Important Compounds of Sodium
- Biological Importance of Sodium and Potassium
- Group 2 Elements - Alkaline Earth Metals
- General Characteristics of the Compounds of the Alkaline Earth Metals
- Anomalous Behaviour of Beryllium
- Some Important Compounds of Calcium
- Biological Importance of Magnesium and Calcium
Some p-Block elements
- Introduction to p-block Elements
- Group 13 Elements - The Boron Family
- Important Trends and Anomalous Properties of Boron
- Important Compounds of Boron
- Preparation and Properties of Aluminium
- Uses of Boron and Aluminium
- Group 14 Elements - The Carbon Family
- Important Trends and Anomalous Behaviour of Carbon
- Straight chains, Branched chains, and Rings of Carbon atoms
- Straight chains, Branched chains, and Rings of Carbon atoms
- Allotropes of Carbon > Diamond
- Allotropes of Carbon > Graphite
- Allotropes of Carbon > Fullerene
- Allotropes of Carbon > Diamond
- Some Important Compounds of Carbon and Silicon - Carbon Monoxide
- Some Important Compounds of Carbon and Silicon - Carbon Dioxide
- Some Important Compounds of Carbon and Silicon - Silicon Tetrachloride
- Some Important Compounds of Carbon and Silicon - Silicon Dioxide
- Some Important Compounds of Carbon and Silicon - Silicones
- Some Important Compounds of Carbon and Silicon - Silicates
- Some Important Compounds of Carbon and Silicon - Zeolites
Environmental Chemistry
- Introduction of Environmental Chemistry
- Atmospheric Pollution
- Tropospheric Pollution - Gaseous Air Pollutants
- Tropospheric Pollution - Particulate Pollutants
- Stratospheric Pollution
- Smog
- Greenhouse Effect and Global Warming
- Industrial Waste
- International Standards for Drinking Water
- Soil Pollution
- Strategies to Control Environmental Pollution
- Green Chemistry - Introduction
- Green Chemistry in Day-to-day Life
Hydrogen
- Position of Hydrogen in the Periodic Table
- Dihydrogen
- Preparation of Dihydrogen
- Properties and Uses of Dihydrogen
- Hydrides
- Structure of Water
- Structure of Ice
- Physical Properties of Water
- Chemical Properties of Water
- Classification of water: Soft and Hard Water
- Causes of Hardness
- Removal of Hardness of Water
- Heavy Water
- Preparation of Hydrogen Peroxide
- Structure of Hydrogen Peroxide
- Physical Properties of Hydrogen Peroxide
- Chemical Properties of Hydrogen Peroxide
- Uses of Hydrogen Peroxide
- Dihydrogen as a Fuel
Aldehydes, Ketones and Carboxylic Acids
- Concepts of Aldehydes, Ketones, and Carboxylic Acids
- Classification of Ketones
- Nature of Carbonyl Group
- Nomenclature of Aldehydes
- Nomenclature of Ketones
- Preparation of Aldehydes
- Preparation of Ketones
- Physical Properties of Aldehydes
- Physical Properties of Ketones
- Chemical Reactions of Aldehydes and Ketones - Nucleophilic Addition Reactions
- Chemical Reactions of Aldehydes and Ketones - Reduction
- Chemical Reactions of Aldehydes and Ketones - Oxidation
- Chemical Reactions of Aldehydes and Ketones - Reactions Due to α-hydrogen
- Chemical Reactions of Aldehydes and Ketones - Other Reactions
- Uses of Aldehydes and Ketones
- Carboxylic Acids
- Classification of Carboxylic Acids
- Nomenclature of Carboxylic Acids
- Structure of the Carboxyl group
- Methods of Preparation of Carboxylic Acids
- Physical Properties of Carboxylic Acids
- Chemical Reactions of Carboxylic Acids - Reactions Involving Cleavege of O-H Bond
- Chemical Reactions of Carboxylic Acids - Reactions Involving Cleavege of C-OH Bond
- Chemical Reactions of Carboxylic Acids - Reactions Involving –COOH Group
- Chemical Reactions of Carboxylic Acids - Substitution Reactions in the Hydrocarbon Part
- Uses of Carboxylic Acids
Alcohols, Phenols and Ethers
- Introduction of Alcohols, Phenols, and Ethers
- Uses of Alcohols
- Classification of Alcohols
- Nomenclature of Alcohols
- Isomerism in Alcohols
- Structure of functional group (-OH) of Alcohols
- Methods of Preparation of Alcohols
- Preparation of Commercially Important Alcohols
- Physical Properties of Alcohols
- Chemical Properties of Alcohols
- Phenols
- Classification of Phenols
- Nomenclature of phenols
- Structure of Functional Group (Ar - OH) of Phenol
- Methods of Preparation of Phenols
- Physical Properties of phenol
- Chemical Properties of Phenol
- Acidic Nature of Phenol
- Uses of Phenols
- Ethers
- Classification of Ethers
- Nomenclature of ethers
- Structure of functional group (R-O-R')
- Isomerism of Ether
- Preparation of Ethers
- Physical Properties of Ethers
- Chemical Properties of Ethers
- Uses of Ethers
Haloalkanes and Haloarenes
- Introduction to Haloalkanes
- Classification of Haloalkanes
- Nomenclature of Haloalkanes and Haloarenes
- Nature of C - X Bond in Haloalkanes
- Methods of Preparation of Haloalkanes
- Physical Properties of Haloalkanes
- Chemical Properties of Haloalkanes
- Stereochemistry
- Nucleophilic substitution Mechanism
- Haloarenes
- Classification of Haloarenes
- Nomenclature of Haloarenes
- Nature of C - X Bond in Haloarenes
- Methods of Preparation of Haloalkanes
- Physical Properties of Haloarenes
- Chemical Properties of Haloarenes
- Polyhalogen Compounds
Surface Chemistry
- Surface Chemistry
- Introduction of Adsorption
- Distinction Between Adsorption and Absorption
- Adsorption
- Mechanism of Adsorption
- Types of Adsorption
- Adsorption Isotherms (Freundlich and Langmuir Adsorption Isotherm)
- Applications of Adsorption
- Factors Affecting Adsorption of Gases on Solids
- Adsorption from Solution Phase
- Catalysis
- Homogeneous and Heterogeneous Catalysis
- Shape-selective Catalysis by Zeolites
- Enzyme Catalysis
- Catalysts in Industry
- Adsorption Theory of Heterogeneous Catalysis
- Catalytic Activity and Selectivity
- Colloids
- Classification of Colloids
- Purification of Colloidal Solution
- Properties of Colloidal Dispersions
- Preparation of Colloids
- Purification of Colloidal Solution
- Properties of Colloidal Solutions
- Colloids Around Us
- Emulsions
Solid State
- States of Matter
- General Characteristics of Solid State
- Amorphous and Crystalline Solids
- Classification of Crystalline Solids
- Bragg's Law and Its Applications
- Crystal Lattices and Unit Cells
- Crystal Lattices and Unit Cells - Primitive and Centred Unit Cells
- Number of Atoms in a Unit Cell
- Close Packed Structures of Solids
- Close Packed Structures - Formula of a Compound and Number of Voids Filled
- Packing Efficiency
- Packing Efficiency in Simple Cubic Lattice
- Efficiency of Packing in Body-centred Cubic Structures
- Packing Efficiency in hcp and ccp Structures
- Calculations Involving Unit Cell Dimensions
- Imperfections in Solids - Introduction
- Imperfections in Solids
- Types of Point Defects - Stoichiometric Defects
- Types of Point Defects - Non-stoichiometric Defects
- Types of Point Defects - Impurity Defects
- Properties of Solids: Electrical Properties
- Properties of Solids: Electrical Properties
- Conduction of Electricity in Metals
- Conduction of Electricity in Semiconductors
- Applications of n-type and p-type Semiconductors
- Properties of Solids: Magnetic Properties
- Solid State
- Band Theory of Metals
Chemistry in Everyday Life
- Chemistry in Everyday Life
- Classification of Drugs
- Designing of a Drug and Action of Drugs on Targets
- Drug-target Interaction - Enzymes as Drug Targets
- Drug-target Interaction - Receptors as Drug Targets
- Therapeutic Action of Different Classes of Drugs - Neurologically Active Drugs
- Antimicrobials
- Therapeutic Action of Different Classes of Drugs - Antifertility Drugs
- Therapeutic Action of Different Classes of Drugs - Antacids
- Therapeutic Action of Different Classes of Drugs - Antihistamines
- Chemicals in Food - Artificial Sweetening Agents and Food Preservatives
- Cleansing Agents
- Preparation from Soap: Saponification
General Principles and Processes of Isolation of Elements
- General Principles and Processes of Isolation of Elements
- Occurrence of Metals
- Metallurgy
- Types of Separation or Concentration of an Ore
- Hydraulic Washing
- Magnetic Separation
- Froth Floatation Method
- Leaching
- Extraction of Crude Metal from Concentrated Ore
- Thermodynamic Principles of Metallurgy
- Application of Thermodynamic Principles of Metallurgy
- Electrochemical Principles of Metallurgy
- Refining of Crude Metals
- Metallurgy of Aluminium
- Basic Principles of Metallurgy > Extraction of Metals
- Refining of Aluminium
- Metallurgy of Copper
- Extraction of Copper
- Occurrence and Principle of Extraction of Zinc from Zinc Blende
- Metallurgy of Iron
- Extraction of Iron
- Uses of Aluminium, Copper, Zinc and Iron
Electrochemistry
- Introduction to Electrochemistry
- Concept of Redox Reactions
- Electrochemical Cells
- Electrodes
- Galvanic or Voltaic Cell
- Electrode Potential and Cell Potential
- Galvanic Cells - Measurement of Electrode Potential
- Electrochemical Series (Electromotive Series)
- Relation Between Gibbs Energy Change and Emf of a Cell
- Nernst Equation
- Nernst Equation
- Equilibrium Constant from Nernst Equation
- Electrochemical Cell and Gibbs Energy of the Reaction
- Conductance of Electrolytic Solutions - Introduction
- Conductance of Electrolytic Solutions
- Measurement of the Conductivity of Ionic Solutions
- Variation of Conductivity and Molar Conductivity with Concentration
- Electrolytic Cells and Electrolysis - Introduction
- Products of Electrolysis
- Batteries
- Primary Batteries
- Secondary Batteries
- Fuel Cells
- Corrosion of Metals
- Prevention of Corrosion
Polymers
- Introduction to Polymers
- Classification of Polymers> Based on Source or Origin
- Classification of Polymers> Based on Structure
- Classification of Polymers> Based on Mode of Polymerisation
- Classification of Polymers> Based on Intermolecular Forces
- Classification Based on the Types of Monomeric Units
- Classification Based on the Biodegradability
- Types of Polymerisation Reactions - Copolymerisation
- Types of Polymerisation Reactions - Addition Polymerisation or Chain Growth Polymerisation
- Types of Polymerisation Reactions - Condensation Polymerisation Or Step Growth Polymerisation
- Some Important Polymers
- Types of Polymerisation Reactions - Rubber
- Molecular Mass of Polymers
- Classification of Polymers> Based on Biodegradability
- Polymers of Commercial Importance
Estimated time: 5 minutes
CBSE: Class 12
Key Points: Tests for Acids, Esters, and Amides
Tests for Acids (Carboxylic acids):
| Test | Observation | Inference |
|---|---|---|
| NaHCO₃ test | Brisk CO₂ effervescence | Presence of –COOH (acid) |
| Fluorescein reaction | Heated with conc. H₂SO₄ + Resorcinol → red solution with intense green fluorescence | Dicarboxylic acid |
| Anhydride formation | White shiny needles on sides of inverted funnel | Dicarboxylic acid |
Distinction: Alcohol vs Phenol vs Carboxylic Acid:
- Alcohol: reacts with Na → H₂ gas; does NOT react with NaOH or NaHCO₃
- Phenol: reacts with Na + NaOH → H₂; does NOT react with NaHCO₃
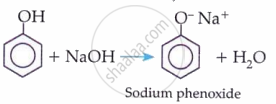
- Carboxylic acid: reacts with Na, NaOH, and NaHCO₃ (gives CO₂)
Tests for Esters:
- \[ \mathrm{RCOOR}' + \mathrm{NaOH} + \text{Phenolphthalein (Pink)} \xrightarrow{\Delta} \mathrm{RCOOH} + \mathrm{R}'\mathrm{OH} \text{ (Colorless solution)} \]
- Esters have sweet/fruity smell
Test for Acid amide:
Acid amides (1°) give smell of ammonia when with alkali.
\[ \mathrm{R} - \overset{\displaystyle \mathrm{O}}{\overset{||}{\mathrm{C}}} - \mathrm{NH}_2 \xrightarrow[\Delta]{\mathrm{NaOH}} \mathrm{R} - \overset{\displaystyle \mathrm{O}}{\overset{||}{\mathrm{C}}} - \mathrm{ONa} + \mathrm{NH}_3 \]
