Topics
Some Basic Concepts of Chemistry
- Importance and Scope of Chemistry
- Historical Approach to Particulate Nature of Matter
- Nature of Matter
- The International System of Units (SI)
- Mass and Weight
- Scientific Notation
- Significant Figures
- Dimensional Analysis
- Law of Conservation of Mass
- Law of Constant Proportions (Law of Definite Proportions)
- Law of Multiple Proportions
- Gay Lussac’s Law of Gaseous Volumes
- Avogadro's Law
- Dalton's Atomic Theory
- Atomic Mass
- Average Atomic Mass
- Molecular Mass
- Formula Mass
- Mole Concept
- Percentage Composition
- Stoichiometry and Stoichiometric Calculations
- Limiting Reagent
- Concentration of a Solution
- Introduction of Some Basic Concepts of Chemistry
Structure of Atom
- Subatomic Particles
- Charge to Mass Ratio of Electron
- Charge on the Electron
- Subatomic Particles
- Atomic Models
- J. J. Thomson’s Atomic Model
- Lord Rutherford’s Atomic model
- Atomic Number (Z) and Mass Number (A)
- Atomic Mass
- Isotopes
- Drawbacks of Rutherford Atomic Model
- Wave Nature of Electromagnetic Radiation
- Electromagnetic Waves : Numericals
- Particle Nature of Electromagnetic Radiation: Planck's Quantum Theory of Radiation
- Evidence for the Quantized Electronic Energy Levels - Atomic Spectra
- Bohr’s Model for Hydrogen Atom
- Dual Behaviour of Matter: De Broglie's relationship
- Heisenberg’s Uncertainty Principle
- Quantum Mechanical Model of Atom
- Quantum Mechanical Model of the Atom - Orbitals and Quantum Numbers
- Quantum Mechanical Model of the Atom - Concept of Shells and Subshells
- Quantum Mechanical Model of the Atom - Shapes of Atomic Orbitals
- Quantum Mechanical Model of the Atom - Energies of Orbitals
- Quantum Mechanical Model of the Atom - Filling of Orbitals in Atom
- Quantum Mechanical Model of the Atom - Electronic Configuration of Atoms
- Quantum Mechanical Model of the Atom - Stability of Completely Filled and Half Filled Subshells
- Structure of Atom Numericals
Classification of Elements and Periodicity in Properties
- Significance of Classification of Elements
- Genesis of Periodic Classification
- Modern Periodic Law and the Present Form of the Periodic Table
- Nomenclature of Elements with Atomic Number Greater than 100
- Periodic Table and Electronic Configuration
- The s-Block Elements
- The p-Block Elements
- The d-Block Elements (Transition Elements)
- The f-Block Elements (Inner-transition Elements)
- Metals, Non-metals and Metalloids
- Atomic Size
- Ionic Radius
- Ionization Enthalpy or Ionization Energy (IE) or Ionization Potential (IP)
- Electron Gain Enthalpy
- Electronegativity
- Periodicity of Valence or Oxidation States
- Anomalous Properties of Second Period Elements
- Periodic Trends and Chemical Reactivity
- Classification of Elements and Periodicity in Properties Numericals
Chemical Bonding and Molecular Structure
- Kossel and Lewis Approach to Chemical Bonding
- Kossel-lewis Approach to Chemical Bonding - Octet Rule
- Kossel-lewis Approach to Chemical Bonding - Covalent Bond
- Lewis Structures (Lewis Representation of Simple Molecules)
- Formal Charge
- Limitations of the Octet Rule
- Ionic or Electrovalent Bond
- Bond Length
- Bond Angle
- Bond Enthalpy
- Bond Order
- Resonance Structures
- Polarity of Bonds
- Valence Shell Electron Pair Repulsion (VSEPR) Theory
- Valence Bond Theory (VBT)
- Valence Bond Theory - Orbital Overlap Concept
- Valence Bond Theory - Directional Properties of Bonds
- Valence Bond Theory - Overlapping of Atomic Orbitals
- Valence Bond Theory - Types of Overlapping and Nature of Covalent Bonds
- Valence Bond Theory - Strength of Sigma (σ) bond and pi (π) bond
- Hybridisation
- Types of Hybridisation
- Hybridisation of Elements Involving d Orbitals
- Molecular Orbital Theory
- Formation of Molecular Orbitals - Linear Combination of Atomic Orbitals (LCAO)
- Conditions for the Combination of Atomic Orbitals
- Types of Molecular Orbitals
- Energy Level Diagram for Molecular Orbitals
- Electronic Configuration and Molecular Behaviour
- Bonding in Some Homonuclear Diatomic Molecules
- Hydrogen Bonding
- Cause of Formation of Hydrogen Bond
- Types of Hydrogen Bonding
- Chemical Bonding and Molecular Structure Numericals
- States of Matter
States of Matter:- Gases and Liquids
- Intermolecular Forces
- Dispersion Forces Or London Forces
- Dipole - Dipole Forces
- Dipole-induced Dipole Forces
- Hydrogen Bond
- Power Plant Based on Thermal Energy
- Intermolecular Forces Vs. Thermal Interactions
- The Gaseous State
- Boyle’s Law (Pressure - Volume Relationship)
- Charles’ Law (Temperature - Volume Relationship)
- Gay Lussac’s Law (Pressure- Temperature Relationship)
- Avogadro's Law
- Density and Molar Mass of a Gaseous Substance
- Dalton’s Law of Partial Pressures
- Kinetic Molecular Theory of Gases
- Behaviour of Real Gases: Deviation from Ideal Gas Behaviour
- Liquefaction of Gases
- Vapour Pressure
- Surface Tension
- Viscosity
- States of Matter
Chemical Thermodynamics
- Thermodynamic Terms
- The State of the System
- The Internal Energy as a State Function - Work
- The Internal Energy as a State Function - Heat
- The Internal Energy as a State Function - the General Case
- Work
- Enthalpy (H)
- Calorimetric Measurement of ΔU and ΔH
- Standard Enthalpy of Reactions
- Enthalpy Changes During Phase Transformations
- Standard Enthalpy of Formation
- Thermochemical Equations
- Hess’ Law of Constant Heat Summation
- Is Decrease in Enthalpy a Criterion for Spontaneity
- Entropy and Spontaneity
- Gibbs Energy and Spontaneity
- Entropy and Second Law of Thermodynamics
- Absolute Entropy and Third Law of Thermodynamics
- Gibbs Energy Change and Equilibrium
Equilibrium
- Concept of Equilibrium
- Solid-liquid Equilibrium
- Liquid-vapour Equilibrium
- Solid - Vapour Equilibrium
- Equilibrium Involving Dissolution of Solid in Liquids
- Equilibrium Involving Dissolution of Gases in Liquids
- General Characteristics of Equilibria Involving Physical Processes
- Equilibrium in Chemical Processes - Dynamic Equilibrium
- Law of Chemical Equilibrium and Equilibrium Constant
- Equilibrium Constant in Gaseous Systems
- Heterogeneous Equlibria
- Predicting the Extent of a Reaction
- Predicting the Direction of the Reaction
- Calculating Equilibrium Concentrations
- Relationship Between Equilibrium Constant K, Reaction Quotient Q and Gibbs Energy G
- Effect of Concentration Change
- Effect of Pressure Change
- Effect of Inert Gas Addition
- Effect of Temperature Change
- Effect of Catalyst
- Ionic Equilibrium in Solution
- Concept of Acid, Base, and Salt
- Arrhenius Concept of Acids and Bases
- Concept of Ionization of Acids and Bases
- The Ionization Constant of Water and Its Ionic Product
- The pH Scale
- Ionization Constants of Weak Acids
- Ionization of Weak Bases
- Relation Between Ka and Kb
- Di- and Polybasic Acids and Di- and Polyacidic Bases
- Factors Affecting Acid Strength
- Common Ion Effect in the Ionization of Acids and Bases
- Hydrolysis of Salts and the Ph of Their Solutions
- Buffer Solutions
- Concept of Solubility Equilibria of Sparingly Soluble Salts
Redox Reactions
- Classical Idea of Redox Reactions - Oxidation and Reduction Reactions
- Redox Reactions in Terms of Electron Transfer Reactions - Introduction
- Redox Reactions in Terms of Electron Transfer Reactions - Competitive Electron Transfer Reactions
- Oxidation Number
- Types of Redox Reactions
- Balancing of Redox Reactions
- Redox Reactions as the Basis for Titrations
- Limitations of Concept of Oxidation Number
- Redox Reactions and Electrode Processes
Organic Chemistry - Some Basic Principles and Techniques
- Tetravalence of Carbon - Shapes of Organic Compounds
- Complete, Condensed and Bond-line Structural Formulas
- Three-dimensional Representation of Organic Molecules
- Classification of Organic Compounds
- Nomenclature
- IUPAC Nomenclature of Alkanes
- Nomenclature of Organic Compounds having Functional Group(s)
- Nomenclature of Substituted Benzene Compounds
- Isomerism
- Fission of a Covalent Bond
- Nucleophiles and Electrophiles
- Electron Movement in Organic Reactions
- Electron Displacement Effects in Covalent Bonds
- Inductive Effect
- Resonance Structure
- Resonance Effect
- Electromeric Effect (E Effect)
- Hyperconjugation
- Types of Organic Reactions and Mechanisms
- Purification of Organic Compounds
- Sublimation
- Crystallisation Method
- Simple Distillation Method
- Solvent Extraction (Using a Separating Funnel Method)
- Chromatography Method
- Qualitative Analysis of Organic Compounds
- Qualitative Analysis of Organic Compounds - Detection of Other Elements
- Quantitative Analysis of Carbon and Hydrogen
- Quantitative Analysis of Nitrogen
- Quantitative Analysis of Halogens
- Quantitative Analysis of Sulphur
- Quantitative Analysis of Phosphorus
- Quantitative Analysis of Oxygen
Hydrocarbons
- Classification of Hydrocarbons
- Alkanes
- Nomenclature and Isomerism
- Preparation of Alkanes from Unsaturated Hydrocarbons, Alkyl Halides and Carboxylic Acids
- Physical Properties of Alkanes
- Chemical Properties of Alkanes
- Conformations (Ethane)
- Alkenes
- Structure of Double Bond (Ethene)
- Nomenclature
- Isomerism
- Preparation of Alkenes from Alkynes, Alkyl Halides, Vicinal Dihalides and Alcohols by Acidic Dehydration
- Physical Properties of Alkenes
- Chemical Properties of Alkenes
- Alkynes
- Nomenclature and Isomerism
- Structure of Triple Bond
- Preparation of Alkynes
- Physical Properties of Alkynes
- Chemical Properties of Alkynes
- Aromatic Hydrocarbons
- Nomenclature and Isomerism
- Structure of Benzene
- Aromaticity (Huckel Rule)
- Preparation of Benzene
- Physical Properties of Aromatic Hydrocarbons
- Chemical Properties of Aromatic Hydrocarbons
- Electrophilic Substitution Reactions
- Concept of Reactivity in Electrophilic Substitution ESR
- Directive Influence of a Functional Group in Monosubstituted Benzene
- Carcinogenicity and Toxicity
Hydrogen
- Position of Hydrogen in the Periodic Table
- Dihydrogen
- Preparation of Dihydrogen
- Properties and Uses of Dihydrogen
- Ionic or Saline Hydrides
- Covalent or Molecular Hydride
- Metallic or Non-stoichiometric (or Interstitial) Hydrides
- Physical Properties of Water
- Structure of Water
- Structure of Ice
- Chemical Properties of Water
- Classification of water: Soft and Hard Water
- Temporary Hardness of Water
- Permanent Hardness of Water
- Preparation of Hydrogen Peroxide
- Physical Properties of Hydrogen Peroxide
- Structure of Hydrogen Peroxide
- Chemical Properties of Hydrogen Peroxide
- Storage of Hydrogen Peroxide
- Uses of Hydrogen Peroxide
- Heavy Water
- Dihydrogen as a Fuel
S-block Elements (Alkali and Alkaline Earth Metals)
- Group 1 Elements - Alkali Metals
- General Characteristics of the Compounds of the Alkali Metals
- Anomalous Properties of Lithium
- Some Important Compounds of Sodium
- Biological Importance of Sodium and Potassium
- Group 2 Elements - Alkaline Earth Metals
- General Characteristics of the Compounds of the Alkaline Earth Metals
- Anomalous Behaviour of Beryllium
- Some Important Compounds of Calcium
- Biological Importance of Magnesium and Calcium
Some P-block Elements
- Introduction to p-block Elements
- Group 13 Elements - The Boron Family
- Important Trends and Anomalous Properties of Boron
- Important Compounds of Boron
- Uses of Boron and Aluminium
- Group 14 Elements - The Carbon Family
- Important Trends and Anomalous Behaviour of Carbon
- Allotropes of Carbon > Diamond
- Allotropes of Carbon > Graphite
- Allotropes of Carbon > Fullerene
- Allotropes of Carbon > Diamond
- Some Important Compounds of Carbon and Silicon - Carbon Monoxide
- Some Important Compounds of Carbon and Silicon - Carbon Dioxide
- Some Important Compounds of Carbon and Silicon - Silicon Dioxide
- Some Important Compounds of Carbon and Silicon - Silicones
- Some Important Compounds of Carbon and Silicon - Silicates
- Some Important Compounds of Carbon and Silicon - Zeolites
Environmental Chemistry
- Tropospheric Pollution - Gaseous Air Pollutants
- Tropospheric Pollution - Particulate Pollutants
- Stratospheric Pollution
- International Standards for Drinking Water
- Soil Pollution
- Industrial Waste
- Strategies to Control Environmental Pollution
- Green Chemistry - Introduction
- Green Chemistry in Day-to-day Life
CISCE: Class 10
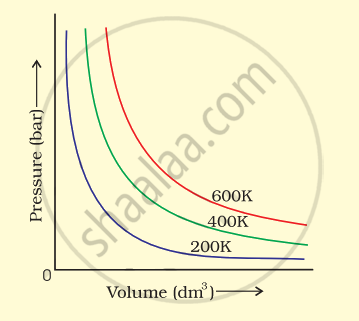
Law: Charles's Law
Charles' Law (Temperature–Volume Relationship)
At constant pressure (P) and number of moles (n), the volume of a gas is directly proportional to its absolute temperature.
\[V\propto T\quad\Rightarrow\quad\frac{V_1}{T_1}=\frac{V_2}{T_2}\]
The V–T curve at constant pressure is called an isobar
Absolute zero = 0 K = –273.15°C — the temperature at which gas volume theoretically becomes zero. It cannot be attained in practice (temperatures of ~0.000001 K have been achieved in labs)
or
Statement:
The volume of a fixed mass of gas is directly proportional to its absolute temperature if the pressure is kept constant.
Mathematically, V ∝ T ⇒ \[\frac {V}{T}\] = constant
Graph: V vs T (Isobar)
 A straight line through the origin when using Kelvin. All lines converge at 0 K (absolute zero).
A straight line through the origin when using Kelvin. All lines converge at 0 K (absolute zero).
Shaalaa.com | Charle's law
Related QuestionsVIEW ALL [4]
Match the following gas laws with the equation representing them.
| (i) Boyle's law | (a) `V oo n` at constant T and p |
| (ii) Charle's law | (b) PTotal = p1 + p2 + p3 + at constant T, V |
| (iii) Dalton's law | (c) `(pV)/T` = Constant T |
| (iv) Avogadro law | (d) `V oo T` at constant n and p |
| (e) `P oo 1/V` at constant n and T |