Topics
Some Basic Concepts of Chemistry
Introduction to Analytical Chemistry
- Introduction of Analytical Chemistry
- Analysis
- Mathematical Operation and Error Analysis
- Determination of Molecular Formula
- Chemical Reactions and Stoichiometric Calculations
- Limiting Reagent
- Concentration of a Solution
- Use of Graph in Analysis
Basic Analytical Techniques
- Introduction of Some Analytical Techniques
- Purification of Solids
- Crystallisation Method
- Fractional Crystallization
- Simple Distillation Method
- Solvent Extraction
- Chromatography Method
- Chromatography Method > Adsorption Chromatography
- Chromatography Method > Partition Chromatography
Structure of Atom
Chemical Bonding
- Concept of Chemical Bonding
- Kossel-lewis Approach to Chemical Bonding - Octet Rule
- Kossel and Lewis Approach to Chemical Bonding
- Formal Charge
- Limitations of the Octet Rule
- Valence Shell Electron Pair Repulsion (VSEPR) Theory
- Valence Bond Theory (VBT)
- Molecular Orbital Theory
- Parameters of Covalent Bond
- Dipole Moment
- Resonance
Redox Reactions
Modern Periodic Table
- Introduction of Periodic Table
- Structure of the Modern Periodic Table
- Periodic Table and Electronic Configuration
- Blockwise Characteristics of Elements
- Periodic Trends in Elemental Properties
Elements of Group 1 and 2
Elements of Group 13, 14 and 15
- Electronic Configuration of Elements of Groups 13, 14 and 15
- Trends in Atomic and Physical Properties of Elements of Groups 13, 14 and 15
- Chemical Properties of the Elements of the Groups 13,14 and 15
- Carbon: A Versatile Element
- Allotropes of Carbon > Diamond
- Molecular Structures of Some Important Compounds of the Group 13, 14 and 15 Elements
- Chemistry of Notable Compounds of Elements of Groups 13, 14 and 15
States of Matter
Adsorption and Colloids
Chemical Equilibrium
- Introduction of Chemical Equilibrium
- Equilibrium in Physical Processes
- Equilibrium in Chemical Processes - Dynamic Equilibrium
- Law of Mass Action and Equilibrium Constant
- Homogeneous Equilibria
- Characteristics of Equilibrium Constant
- Applications of Equilibrium Constants
- Le Chaterlier's Principle and Factors Altering the Composition of Equilibrium
- Industrial Application
Nuclear Chemistry and Radioactivity
- Introduction: Nuclear Chemistry is a Branch of Physical Chemistry
- Classification of Nuclides
- Nuclear Stability
- Radioactivity
- Radioactive Decays
- Modes of Decay
- Nuclear Reactions
- Applications of Radio Isotopes
Basic Principles of Organic Chemistry
- Organic Chemistry
- Structural Representation of Organic Molecules
- Classification of Organic Compounds
- Nomenclature
- Isomerism
- Theoretical Basis of Organic Reactions
Hydrocarbons
Chemistry in Everyday Life
Estimated time: 8 minutes
CBSE: Class 12
Maharashtra State Board: Class 12
Maharashtra State Board: Class 12
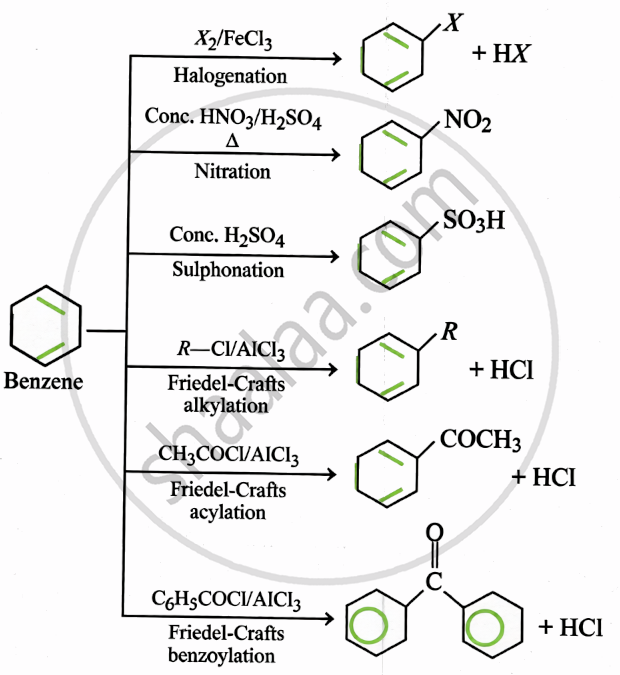
Key Points: Aromatic Hydrocarbons
Cyclic, planar hydrocarbons with delocalised π electrons.
Benzenoids contain benzene ring; Non-benzenoids are aromatic without benzene ring.
Structure of Benzene:
- All 6 C atoms are sp² hybridised
- Unhybridised p-orbitals overlap laterally → delocalised π bonds
- Bond length = 139 pm (uniform, due to resonance)
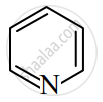
Huckel's Rule
- Cyclic and planar
- Each ring atom has a p-orbital
- Contains (4n + 2) π electrons (n = 0, 1, 2…)
Examples: Benzene, Naphthalene, Pyridine.
Physical Properties:
- Colourless liquid, sweet smell
- BP = 353 K, MP = 278.5 K
- Immiscible with water; burns with sooty flame
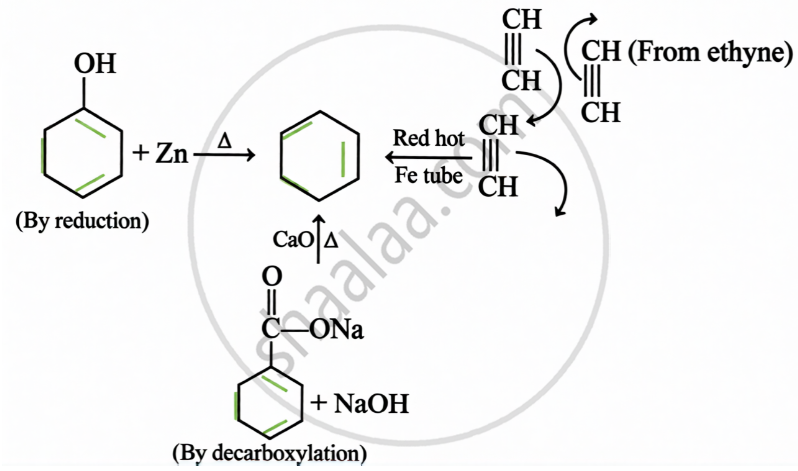
Preparation:
Electrophilic Substitution: