Advertisements
Advertisements
प्रश्न
Read the following reaction and answer the questions given below:

- Write the name of the reaction.
- Identify the electrophile in it.
- How is this electrophile generated?
Advertisements
उत्तर
- The name of the reaction is Friedel-Craft’s alkylation reaction.
- The electrophile in the reaction is +CH3.
- The electrophile +CH3 is generated as follows:
\[\ce{\underset{\text{Methyl chloride}}{CH3 -}Cl + AlCl3 -> \underset{\text{Electrophile}}{^+CH3 +}AlCl^-_4}\]
APPEARS IN
संबंधित प्रश्न
Name the following:
The hydrocarbon said to possess carcinogenic property.
Identify the main product of the reaction:
\[\ce{HC ≡ C - CH3 ->[H2][Pd-C/quinoline]}\] _______.
Identify the main product of the reaction:
\[\ce{H - C ≡ C - H + H - O ->[{40%} H2SO4][{1%} HgSO4]}\] _____.
Name two reagents used for acylation of benzene.
Which of the following compounds will not undergo Friedal – crafts reaction easily ? (NEET)
Give IUPAC name for the following compound.
Ethyl isopropyl acetylene
How is propyne prepared from an alkylene dihalide ?
Describe the mechanism of Nitration of benzene.
Suggest a simple chemical test to distinguish propane and propene.
Suggest the route for the preparation of the following from benzene.
m - dinitro benzene
How will you convert ethyl chloride into n – butane?
What happens when ethylene is passed through cold dilute alkaline potassium permanganate.
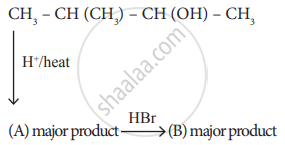
Identify A and B.
How will distinguish 1 – butyne and 2 – butyne?
Which of the following is an aromatic compound?
Which of the following is NOT a hetero-aromatic compound?
A compound with molecular formula C4H4O has all the four carbon atom and the oxygen atom in the ring. It also has two carbon-carbon double bonds. The compound is ____________.
Phenol on distillation with zinc dust gives ____________.
Which of the following is INCORRECT for aromatic hydrocarbons?
Direct bromination of benzene with excess reagent results in the formation of ____________.
Conversion of hexane into benzene involves the reaction of ______.
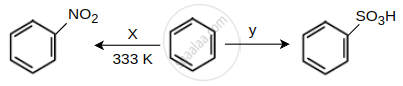
Identify the X and Y in the following reactions.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..............................}\ce{CH3}\\
\phantom{...........................}|\\
\ce{CH3 - C = CH2 + HBr ->[Benzoyl][peroxide] H3C - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{.................................}
\end{array}\]
- Write the IUPAC name of the product.
- State the rule that governs the formation of this product.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{.............................}\ce{CH3}\\
\phantom{...........................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide] CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{.................................}\\
\end{array}\]
- Write IUPAC name of the product.
- State the rule that governs formation of this product.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..............................}\ce{CH3}\\
\phantom{...........................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide] CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{.................................}
\end{array}\]
- Write the IUPAC name of the product.
- State the rule that governs the formation of this product.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..............................}\ce{CH3}\\
\phantom{............................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide]CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{.................................}
\end{array}\]
- Write IUPAC name of the product.
- State the rule that governs formation of this product.
Which of the following is most stable?
