Advertisements
Advertisements
प्रश्न
Identify A, B, C in the following reaction sequence:
\[\ce{CH3 - CH = CH2 ->[Br2/CCl4][room temperature] A ->[Zn] B ->[dil. alkaline][KMnO4] C}\]
Advertisements
उत्तर
\[\begin{array}{cc}
\ce{H\phantom{....}H\phantom{...............}}\\
|\phantom{.....}|\phantom{...............}\\
\ce{CH3\underset{Propene}{- CH =} CH2 ->[Br2/CCl4][room temperature] CH3 - C - C - H ->[Zn] CH3 \underset{\overset{Propene}{(B)}}{- CH = C}H2 ->[dil.alkaline][KMnO4] CH3 - CH - CH2}\\
\phantom{........................................}|\phantom{.....}|\phantom{..............................................}|\phantom{......}|\phantom{..}\\
\ce{\phantom{.......................................}\underset{\overset{1,2-Dibromopropane}{(A)}}{Br\phantom{...} Br}\phantom{...........................................}\underset{\overset{Propane-1,2-diol}{(C)}}{OH\phantom{...}OH}}\end{array}\]
APPEARS IN
संबंधित प्रश्न
Name the following:
The hydrocarbon said to possess carcinogenic property.
Write the balanced chemical reaction to get benzene from Phenol.
Predict the possible product of the following reaction.
sulphonation of chlorobenzene
Identify the main product of the reaction:
\[\ce{H - C ≡ C - H + H - O ->[{40%} H2SO4][{1%} HgSO4]}\] _____.
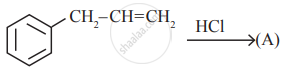 is
is
An alkane is obtained by decarboxylation of sodium propionate. Same alkane can be prepared by ______.
Which of the following is aliphatic saturated hydrocarbon?
Identify the compound 'Z' in the following reaction
\[\ce{C2H6O ->[Al2O3][623 K] X ->[O3] Y ->[Zn/H2O] (Z)}\]
Identify the compound A, B, C and D in the following series of reactions.
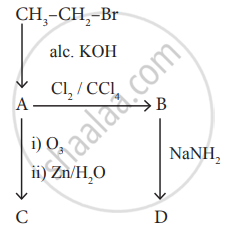
How is propyne prepared from an alkylene dihalide ?
Write the chemical equations for combustion of propane.
How will you convert ethyl chloride into n – butane?
What happens when ethylene is passed through cold dilute alkaline potassium permanganate.
Write the structure of the following alkanes.
2, 3 – Dimethyl – 6 – (2 – methyl propyl) decane
How will you prepare propane from a sodium salt of fatty acid?
−Cl group is ____________.
Phenol on distillation with zinc dust gives ____________.
Which of the following is NOT alicyclic compound?
The alkane formed on heating sodium butanoate with sodalime is ______.
Dow's process is used for the synthesis of an aromatic compound (X). Identify X.
Which of the following molecules has shortest C - C bond length?
Which of the following reagents can bring about following conversion?
\[\ce{But-1-ene -> Butan-2-ol}\]
Which of the following compounds on bromination yields ![]() ?
?
Arenes on treatments with chlorine in the presence of ferric chloride as a catalyst undergo what type of reaction?
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..............................}\ce{CH3}\\
\phantom{...........................}|\\
\ce{CH3 - C = CH2 + HBr->[Benzoyl][peroxide] H3C - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{.................................}
\end{array}\]
- Write the IUPAC name of the product.
- State the rule that governs the formation of this product.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..............................}\ce{CH3}\\
\phantom{...........................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide] CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{.................................}
\end{array}\]
- Write the IUPAC name of the product.
- State the rule that governs the formation of this product.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{...............................}\ce{CH3}\\
\phantom{............................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide]CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{..................................}
\end{array}\]
- Write the IUPAC name of the product.
- State the rule that governs the formation of this product.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..............................}\ce{CH3}\\
\phantom{............................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide]CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{.................................}
\end{array}\]
- Write IUPAC name of the product.
- State the rule that governs formation of this product.
Which of the following is most stable?
