Advertisements
Advertisements
प्रश्न
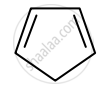
Identify giving reason whether the following compound is aromatic or not.
Advertisements
उत्तर

The compound is non-aromatic since it has 4π electrons and hence, does not obey the Huckel rule of aromaticity.
APPEARS IN
संबंधित प्रश्न
Identify the main product of the reaction.
\[\begin{array}{cc}
\ce{CH3 - CH - CH3 ->[{60%}H2SO4][373 K]}\\
|\phantom{............}\\\ce{OH}\phantom{..........}
\end{array}\] _______.
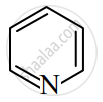
Identify giving reason whether the following compound is aromatic or not.
Name two reagents used for acylation of benzene.
Read the following reaction and answer the questions given below:

- Write the name of the reaction.
- Identify the electrophile in it.
- How is this electrophile generated?
Major product of the below mentioned reaction is, \[\ce{(CH3 )2 C = CH2 ->[ICI]}\]
Which one of the following is non aromatic?
Some meta-directing substituents in aromatic substitution are given. Which one is most deactivating?
Which of the following is aliphatic saturated hydrocarbon?
Identify the compound 'Z' in the following reaction
\[\ce{C2H6O ->[Al2O3][623 K] X ->[O3] Y ->[Zn/H2O] (Z)}\]
Give IUPAC name for the following compound.
\[\ce{CH ≡ C – C ≡ C – C ≡ CH}\]
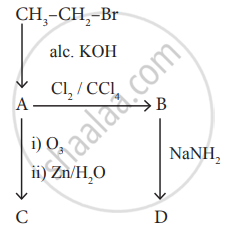
Identify the compound A, B, C and D in the following series of reactions.
Write short notes on ortho, para directors in aromatic electrophilic substitution reactions.
How is propyne prepared from an alkylene dihalide ?
Suggest a simple chemical test to distinguish propane and propene.
How does Huckel rule help to decide the aromatic character of a compound?
Suggest the route for the preparation of the following from benzene.
4 – chlorotoluene
What happens when ethylene is passed through cold dilute alkaline potassium permanganate.
How will you prepare propane from a sodium salt of fatty acid?
Which among the following compounds has highest boiling point?
Phenol on distillation with zinc dust gives ____________.
Direct bromination of benzene with excess reagent results in the formation of ____________.
Sodium benzoate on decarboxylation gives ____________.
α-butylene when subjected to hydroboration oxidation reaction, yields ____________.
Which of the following compounds on bromination yields ![]() ?
?
Arenes on treatments with chlorine in the presence of ferric chloride as a catalyst undergo what type of reaction?
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..............................}\ce{CH3}\\
\phantom{...........................}|\\
\ce{CH3 - C = CH2 + HBr->[Benzoyl][peroxide] H3C - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{.................................}
\end{array}\]
- Write the IUPAC name of the product.
- State the rule that governs the formation of this product.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..............................}\ce{CH3}\\
\phantom{...........................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide]CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{.................................}\\
\end{array}\]
- Write the IUPAC name of the product.
- State the rule that governs the formation of this product.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..............................}\ce{CH3}\\
\phantom{............................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide]CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{.................................}
\end{array}\]
- Write IUPAC name of the product.
- State the rule that governs formation of this product.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..........................}\ce{CH3}\\\phantom{........................}|\\\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide] CH3 - CH - CH2Br}\\
|\phantom{......................................}\\
\ce{CH3}\phantom{....................................}\end{array}\]
- Write the IUPAC name of the product.
- State the rule that governs the formation of this product.
