Advertisements
Advertisements
प्रश्न
Suggest a simple chemical test to distinguish propane and propene.
Advertisements
उत्तर
Propene decolourises Br2/H2O it forms dibromo compound but propane does not react with Br2/ H2O.
APPEARS IN
संबंधित प्रश्न
Identify the main product of the reaction.
\[\begin{array}{cc}
\ce{CH3 - CH - CH3 ->[{60%}H2SO4][373 K]}\\
|\phantom{............}\\\ce{OH}\phantom{..........}
\end{array}\] _______.
Identify A, B, C in the following reaction sequence:
\[\ce{CH3 - CH = CH2 ->[Br2/CCl4][room temperature] A ->[Zn] B ->[dil. alkaline][KMnO4] C}\]
Give IUPAC name for the following compound.
\[\ce{CH ≡ C – C ≡ C – C ≡ CH}\]
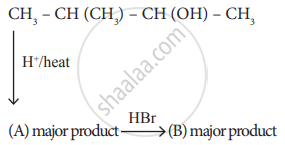
Identify A and B.
How will distinguish 1 – butyne and 2 – butyne?
Which of the following is INCORRECT for aromatic hydrocarbons?
Direct bromination of benzene with excess reagent results in the formation of ____________.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..............................}\ce{CH3}\\
\phantom{............................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide] CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH_3}\phantom{.................................}
\end{array}\]
- Write the IUPAC name of the product.
- State the rule that governs the formation of this product.
Identify ortho and para-directing groups from the following:
\[\ce{\underset{I}{–CHO},\underset{II}{–NHCOCH3},\underset{III}{–OCH3},\underset{IV}{–SO3H}}\]
Benzene reacts with CH3COCl + AICl3 to give ______.
