Advertisements
Advertisements
Question
Suggest a simple chemical test to distinguish propane and propene.
Advertisements
Solution
Propene decolourises Br2/H2O it forms dibromo compound but propane does not react with Br2/ H2O.
APPEARS IN
RELATED QUESTIONS
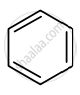
Identify giving reason whether the following compound is aromatic or not.
An alkane is obtained by decarboxylation of sodium propionate. Same alkane can be prepared by ______.
Give IUPAC name for the following compound.
\[\ce{CH ≡ C – C ≡ C – C ≡ CH}\]
Suggest the route for the preparation of the following from benzene.
4 – chlorotoluene
Write the structure of the following alkanes.
2, 3 – Dimethyl – 6 – (2 – methyl propyl) decane
How will distinguish 1 – butyne and 2 – butyne?
α-butylene when subjected to hydroboration oxidation reaction, yields ____________.
Which of the following molecules has shortest C - C bond length?
Which of the following compounds on bromination yields ![]() ?
?
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..........................}\ce{CH3}\\\phantom{........................}|\\\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide] CH3 - CH - CH2Br}\\
|\phantom{......................................}\\
\ce{CH3}\phantom{....................................}\end{array}\]
- Write the IUPAC name of the product.
- State the rule that governs the formation of this product.
