Advertisements
Advertisements
Question
An alkane is obtained by decarboxylation of sodium propionate. Same alkane can be prepared by ______.
Options
Catalytic hydrogenation of propene
action of sodium metal on iodomethane
reduction of 1 – chloro propane
reduction of bromomethane
Advertisements
Solution
An alkane is obtained by decarboxylation of sodium propionate. Same alkane can be prepared by action of sodium metal on iodomethane.
APPEARS IN
RELATED QUESTIONS
Name the following:
The hydrocarbon said to possess carcinogenic property.
Predict the possible product of the following reaction.
Chlorination of nitrobenzene
Identify the main product of the reaction:
\[\ce{HC ≡ C - CH3 ->[H2][Pd-C/quinoline]}\] _______.
Identify the main product of the reaction:
\[\ce{H - C ≡ C - H + H - O ->[{40%} H2SO4][{1%} HgSO4]}\] _____.
Which of the following can be used as the halide component for friedal - crafts reaction?
Direct bromination of benzene with excess reagent results in the formation of ____________.
Dow's process is used for the synthesis of an aromatic compound (X). Identify X.
The treatment of benzene with iso - butene in the presence of sulphuric acid gives:
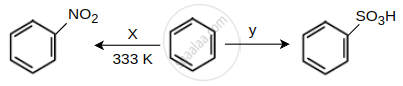
Identify the X and Y in the following reactions.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..............................}\ce{CH3}\\
\phantom{............................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide]CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{.................................}
\end{array}\]
- Write IUPAC name of the product.
- State the rule that governs formation of this product.
