Advertisements
Advertisements
Question
Identify the main product of the reaction:
\[\ce{HC ≡ C - CH3 ->[H2][Pd-C/quinoline]}\] _______.
Advertisements
Solution
\[\ce{HC \underset{\text{Propyne}}{≡ C - }CH3 ->[H2][Pd-C/quinoline] H2C \underset{\text{Propene}}{= CH - CH3}}\]
APPEARS IN
RELATED QUESTIONS
Predict the possible product of the following reaction.
Chlorination of nitrobenzene
Predict the possible product of the following reaction.
bromination of phenol
Identify giving reason whether the following compound is aromatic or not.
Which one of the following is non aromatic?
Which of the following compounds will not undergo Friedal – crafts reaction easily ? (NEET)
Identify the compound 'Z' in the following reaction
\[\ce{C2H6O ->[Al2O3][623 K] X ->[O3] Y ->[Zn/H2O] (Z)}\]
Give IUPAC name for the following compound.
Ethyl isopropyl acetylene
How is propyne prepared from an alkylene dihalide ?
Suggest a simple chemical test to distinguish propane and propene.
Write the chemical equations for combustion of propane.
How does Huckel rule help to decide the aromatic character of a compound?
Suggest the route for the preparation of the following from benzene.
4 – chlorotoluene
Suggest the route for the preparation of the following from benzene.
Bromo benzene
What happens when Isobutylene is treated with acidified potassium permanganate?
How will you convert ethyl chloride into ethane?
What happens when ethylene is passed through cold dilute alkaline potassium permanganate.
Write the structure of the following alkanes.
5 – (2 – Ethyl butyl) – 3, 3 – dimethyldecane
Phenol on distillation with zinc dust gives ____________.
According to Huckel rule, a cyclic π molecular orbital formed by overlap of p orbitals must contain ____________ p electrons.
α-butylene when subjected to hydroboration oxidation reaction, yields ____________.
Which of the following molecules has shortest C - C bond length?
Conversion of hexane into benzene involves the reaction of ______.
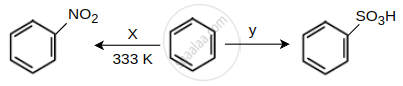
Identify the X and Y in the following reactions.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..............................}\ce{CH3}\\
\phantom{...........................}|\\
\ce{CH3 - C = CH2 + HBr->[Benzoyl][peroxide] H3C - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{.................................}
\end{array}\]
- Write the IUPAC name of the product.
- State the rule that governs the formation of this product.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..............................}\ce{CH3}\\
\phantom{............................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide] CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH_3}\phantom{.................................}
\end{array}\]
- Write the IUPAC name of the product.
- State the rule that governs the formation of this product.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..............................}\ce{CH3}\\
\phantom{...........................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide] CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{.................................}
\end{array}\]
- Write the IUPAC name of the product.
- State the rule that governs the formation of this product.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..............................}\ce{CH3}\\
\phantom{............................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide] CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{..................................}
\end{array}\]
- Write IUPAC name of the product.
- State the rule that governs formation of this product.
