Advertisements
Advertisements
प्रश्न
Identify the main product of the reaction:
\[\ce{HC ≡ C - CH3 ->[H2][Pd-C/quinoline]}\] _______.
Advertisements
उत्तर
\[\ce{HC \underset{\text{Propyne}}{≡ C - }CH3 ->[H2][Pd-C/quinoline] H2C \underset{\text{Propene}}{= CH - CH3}}\]
APPEARS IN
संबंधित प्रश्न
Name the following:
The hydrocarbon said to possess carcinogenic property.
Write the balanced chemical reaction to get benzene from Sodium benzoate.
Predict the possible product of the following reaction.
bromination of phenol
Identify the main product of the reaction.
\[\begin{array}{cc}
\ce{CH3 - CH - CH3 ->[{60%}H2SO4][373 K]}\\
|\phantom{............}\\\ce{OH}\phantom{..........}
\end{array}\] _______.
Identify the main product of the reaction:
\[\ce{H - C ≡ C - H + H - O ->[{40%} H2SO4][{1%} HgSO4]}\] _____.
Identify giving reason whether the following compound is aromatic or not.
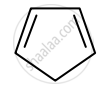
Identify giving reason whether the following compound is aromatic or not.

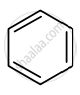
Identify giving reason whether the following compound is aromatic or not.
Name two reagents used for acylation of benzene.
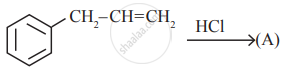 is
is
Which of the following is aliphatic saturated hydrocarbon?
2 – butyne on chlorination gives ______.
Describe the mechanism of Nitration of benzene.
Write the chemical equations for combustion of propane.
How does Huckel rule help to decide the aromatic character of a compound?
Suggest the route for the preparation of the following from benzene.
m - dinitro benzene
Which of the following is NOT a hetero-aromatic compound?
Identify the hydrocarbon compound from following containing carbon atoms in the range of C6 to C8?
A compound with molecular formula C4H4O has all the four carbon atom and the oxygen atom in the ring. It also has two carbon-carbon double bonds. The compound is ____________.
−Cl group is ____________.
The alkane formed on heating sodium butanoate with sodalime is ______.
Arenes on treatments with chlorine in the presence of ferric chloride as a catalyst undergo what type of reaction?
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..............................}\ce{CH3}\\
\phantom{...........................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide]CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{.................................}\\
\end{array}\]
- Write the IUPAC name of the product.
- State the rule that governs the formation of this product.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{.............................}\ce{CH3}\\
\phantom{...........................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide] CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{.................................}\\
\end{array}\]
- Write IUPAC name of the product.
- State the rule that governs formation of this product.
Which of the following statement is NOТ correct for aromatic compounds?
