Advertisements
Advertisements
प्रश्न
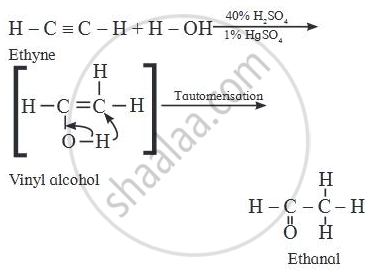
Identify the main product of the reaction:
\[\ce{H - C ≡ C - H + H - O ->[{40%} H2SO4][{1%} HgSO4]}\] _____.
Advertisements
उत्तर
Alkynes react with water in the presence of 40% sulphuric acid and 1% mercuric sulphate to form aldehydes or ketones i.e. carbonyl compounds.
Notes
In the textbook question is wrong.
APPEARS IN
संबंधित प्रश्न
Write the balanced chemical reaction to get benzene from Sodium benzoate.
Identify the main product of the reaction.
\[\begin{array}{cc}
\ce{CH3 - CH - CH3 ->[{60%}H2SO4][373 K]}\\
|\phantom{............}\\\ce{OH}\phantom{..........}
\end{array}\] _______.
Identify the main product of the reaction:
\[\ce{HC ≡ C - CH3 ->[H2][Pd-C/quinoline]}\] _______.
Identify giving reason whether the following compound is aromatic or not.
Identify giving reason whether the following compound is aromatic or not.

Read the following reaction and answer the questions given below:

- Write the name of the reaction.
- Identify the electrophile in it.
- How is this electrophile generated?
Which one of the following is non aromatic?
Which of the following compounds will not undergo Friedal – crafts reaction easily ? (NEET)
An alkane is obtained by decarboxylation of sodium propionate. Same alkane can be prepared by ______.
Identify the compound 'Z' in the following reaction
\[\ce{C2H6O ->[Al2O3][623 K] X ->[O3] Y ->[Zn/H2O] (Z)}\]
Give IUPAC name for the following compound.
Ethyl isopropyl acetylene
Give IUPAC name for the following compound.
\[\ce{CH ≡ C – C ≡ C – C ≡ CH}\]
Write short notes on ortho, para directors in aromatic electrophilic substitution reactions.
How does Huckel rule help to decide the aromatic character of a compound?
Suggest the route for the preparation of the following from benzene.
3 – chloro nitrobenzene
How will you convert ethyl chloride into n – butane?
Write the structure of the following alkanes.
2, 3 – Dimethyl – 6 – (2 – methyl propyl) decane
How will you prepare propane from a sodium salt of fatty acid?
Complete the following:
\[\begin{array}{cc}
\ce{CH2 - CH2 ->[Zn/C2H5OH]}\\
|\phantom{.......}|\phantom{..............}\\
\ce{Br}\phantom{.....}\ce{Br}\phantom{..............}
\end{array}\]
−Cl group is ____________.
Which of the following is INCORRECT for aromatic hydrocarbons?
Which of the following compounds is NOT obtained in thermal decomposition of propane in absence of air?
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..............................}\ce{CH3}\\
\phantom{...........................}|\\
\ce{CH3 - C = CH2 + HBr ->[Benzoyl][peroxide] H3C - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{.................................}
\end{array}\]
- Write the IUPAC name of the product.
- State the rule that governs the formation of this product.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..............................}\ce{CH3}\\
\phantom{............................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide] CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{..................................}
\end{array}\]
- Write IUPAC name of the product.
- State the rule that governs formation of this product.
Which of the following statement is NOТ correct for aromatic compounds?
Identify 'A' in the following reaction.
\[\ce{\underset{}{A + Chloromethane} ->[anhydrous][AlCl3] \underset{\underset{}{}}{2-Chlorotoluene + 4-Chlorotoluene}}\]
Benzene reacts with CH3COCl + AICl3 to give ______.
