Advertisements
Advertisements
Question
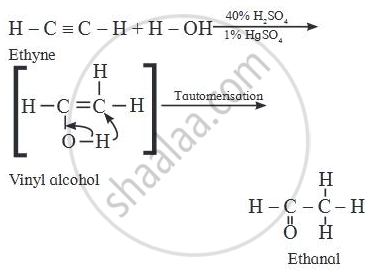
Identify the main product of the reaction:
\[\ce{H - C ≡ C - H + H - O ->[{40%} H2SO4][{1%} HgSO4]}\] _____.
Advertisements
Solution
Alkynes react with water in the presence of 40% sulphuric acid and 1% mercuric sulphate to form aldehydes or ketones i.e. carbonyl compounds.
Notes
In the textbook question is wrong.
APPEARS IN
RELATED QUESTIONS
Write the balanced chemical reaction to get benzene from Sodium benzoate.
Predict the possible product of the following reaction.
Chlorination of nitrobenzene
Predict the possible product of the following reaction.
sulphonation of chlorobenzene
Identify the main product of the reaction:
\[\ce{HC ≡ C - CH3 ->[H2][Pd-C/quinoline]}\] _______.
Identify giving reason whether the following compound is aromatic or not.

Identify giving reason whether the following compound is aromatic or not.
Read the following reaction and answer the questions given below:

- Write the name of the reaction.
- Identify the electrophile in it.
- How is this electrophile generated?
Major product of the below mentioned reaction is, \[\ce{(CH3 )2 C = CH2 ->[ICI]}\]
Some meta-directing substituents in aromatic substitution are given. Which one is most deactivating?
2 – butyne on chlorination gives ______.
How is propyne prepared from an alkylene dihalide ?
Suggest the route for the preparation of the following from benzene.
4 – chlorotoluene
What happens when Isobutylene is treated with acidified potassium permanganate?
How will you prepare propane from a sodium salt of fatty acid?
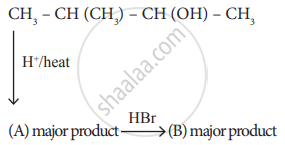
Identify A and B.
Complete the following:
\[\begin{array}{cc}
\ce{CH2 - CH2 ->[Zn/C2H5OH]}\\
|\phantom{.......}|\phantom{..............}\\
\ce{Br}\phantom{.....}\ce{Br}\phantom{..............}
\end{array}\]
How will distinguish 1 – butyne and 2 – butyne?
A compound with molecular formula C4H4O has all the four carbon atom and the oxygen atom in the ring. It also has two carbon-carbon double bonds. The compound is ____________.
−Cl group is ____________.
The alkane formed on heating sodium butanoate with sodalime is ______.
Dow's process is used for the synthesis of an aromatic compound (X). Identify X.
Which of the following molecules has shortest C - C bond length?
Which of the following reagents can bring about following conversion?
\[\ce{But-1-ene -> Butan-2-ol}\]
Conversion of hexane into benzene involves the reaction of ______.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..............................}\ce{CH3}\\
\phantom{...........................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide]CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{.................................}\\
\end{array}\]
- Write the IUPAC name of the product.
- State the rule that governs the formation of this product.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{.............................}\ce{CH3}\\
\phantom{...........................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide] CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{.................................}\\
\end{array}\]
- Write IUPAC name of the product.
- State the rule that governs formation of this product.
Identify 'A' in the following reaction.
\[\ce{\underset{}{A + Chloromethane} ->[anhydrous][AlCl3] \underset{\underset{}{}}{2-Chlorotoluene + 4-Chlorotoluene}}\]
Identify ortho and para-directing groups from the following:
\[\ce{\underset{I}{–CHO},\underset{II}{–NHCOCH3},\underset{III}{–OCH3},\underset{IV}{–SO3H}}\]
Benzene reacts with CH3COCl + AICl3 to give ______.
