Advertisements
Advertisements
Question
Major product of the below mentioned reaction is, \[\ce{(CH3 )2 C = CH2 ->[ICI]}\]
Options
2 – chloro – 1 – iode – 2 – methyl propane
1 – chloro – 2 – iodo – 2 – methyl propane
1, 2 – dichloro – 2 – methyl propane
1, 2 – diiodo – 2 – methyl propane
Advertisements
Solution
2 – chloro – 1 – iode – 2 – methyl propane
APPEARS IN
RELATED QUESTIONS
Identify the main product of the reaction:
\[\ce{HC ≡ C - CH3 ->[H2][Pd-C/quinoline]}\] _______.
Identify giving reason whether the following compound is aromatic or not.
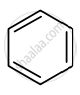
Identify the compound 'Z' in the following reaction
\[\ce{C2H6O ->[Al2O3][623 K] X ->[O3] Y ->[Zn/H2O] (Z)}\]
2 – butyne on chlorination gives ______.
Write short notes on ortho, para directors in aromatic electrophilic substitution reactions.
Write the chemical equations for combustion of propane.
How will you prepare propane from a sodium salt of fatty acid?
α-butylene when subjected to hydroboration oxidation reaction, yields ____________.
Which of the following compounds is NOT obtained in thermal decomposition of propane in absence of air?
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..............................}\ce{CH3}\\
\phantom{...........................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide]CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{.................................}\\
\end{array}\]
- Write the IUPAC name of the product.
- State the rule that governs the formation of this product.
