Advertisements
Advertisements
Question
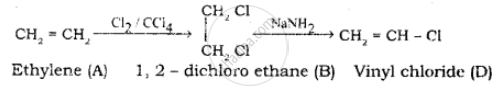
Identify the compound A, B, C and D in the following series of reactions.
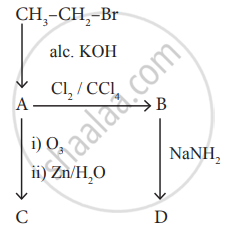
Advertisements
Solution
\[\ce{\underset{\text{Ethyl bromide}}{CH3 - CH2 - Br} ->[alc.KOH] \underset{\text{Ethylene (A)}}{CH2=CH2}}\]

APPEARS IN
RELATED QUESTIONS
Predict the possible product of the following reaction.
Chlorination of nitrobenzene
Identify A, B, C in the following reaction sequence:
\[\ce{CH3 - CH = CH2 ->[Br2/CCl4][room temperature] A ->[Zn] B ->[dil. alkaline][KMnO4] C}\]
Which one of the following is non aromatic?
Which of the following compounds will not undergo Friedal – crafts reaction easily ? (NEET)
Suggest a simple chemical test to distinguish propane and propene.
Which of the following is NOT alicyclic compound?
Dow's process is used for the synthesis of an aromatic compound (X). Identify X.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{...............................}\ce{CH3}\\
\phantom{............................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide]CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{..................................}
\end{array}\]
- Write the IUPAC name of the product.
- State the rule that governs the formation of this product.
Which of the following statement is NOТ correct for aromatic compounds?
Identify 'A' in the following reaction.
\[\ce{\underset{}{A + Chloromethane} ->[anhydrous][AlCl3] \underset{\underset{}{}}{2-Chlorotoluene + 4-Chlorotoluene}}\]
