Advertisements
Advertisements
प्रश्न
How will you convert ethyl chloride into n – butane?
Advertisements
उत्तर
\[\ce{\underset{\text{}Ethyl chloride}{C2H5Cl + 2Na +}Cl - C2H5 ->[-2NaBr][dry ether] CH3 - \underset{\text{n - butane}}{CH2 - CH2 - CH3}}\]
APPEARS IN
संबंधित प्रश्न
Identify giving reason whether the following compound is aromatic or not.
Read the following reaction and answer the questions given below:

- Write the name of the reaction.
- Identify the electrophile in it.
- How is this electrophile generated?
Identify the compound (A) in the following reaction
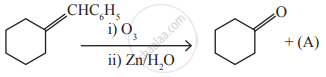
Which one of the following is non aromatic?
Which of the following can be used as the halide component for friedal - crafts reaction?
Suggest the route for the preparation of the following from benzene.
3 – chloro nitrobenzene
What happens when Isobutylene is treated with acidified potassium permanganate?
How will you prepare propane from a sodium salt of fatty acid?
Which of the following is NOT a hetero-aromatic compound?
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..............................}\ce{CH3}\\
\phantom{...........................}|\\
\ce{CH3 - C = CH2 + HBr->[Benzoyl][peroxide] H3C - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{.................................}
\end{array}\]
- Write the IUPAC name of the product.
- State the rule that governs the formation of this product.
