Advertisements
Advertisements
Question
How will you convert ethyl chloride into n – butane?
Advertisements
Solution
\[\ce{\underset{\text{}Ethyl chloride}{C2H5Cl + 2Na +}Cl - C2H5 ->[-2NaBr][dry ether] CH3 - \underset{\text{n - butane}}{CH2 - CH2 - CH3}}\]
APPEARS IN
RELATED QUESTIONS
Write the balanced chemical reaction to get benzene from Phenol.
Consider the nitration of benzene using mixed con H2SO4 and HNO3 if a large quantity of KHSO4 is added to the mixture, the rate of nitration will be ______.
2 – butyne on chlorination gives ______.
Identify the compound A, B, C and D in the following series of reactions.
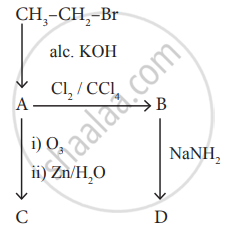
Suggest the route for the preparation of the following from benzene.
m - dinitro benzene
How will you convert ethyl chloride into ethane?
A compound with molecular formula C4H4O has all the four carbon atom and the oxygen atom in the ring. It also has two carbon-carbon double bonds. The compound is ____________.
The alkane formed on heating sodium butanoate with sodalime is ______.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..............................}\ce{CH3}\\
\phantom{............................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide]CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{.................................}
\end{array}\]
- Write IUPAC name of the product.
- State the rule that governs formation of this product.
Identify ortho and para-directing groups from the following:
\[\ce{\underset{I}{–CHO},\underset{II}{–NHCOCH3},\underset{III}{–OCH3},\underset{IV}{–SO3H}}\]
