Advertisements
Advertisements
प्रश्न
Describe the conformers of n – butane.
Advertisements
उत्तर
- Conformations of n-Butane:
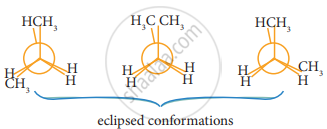
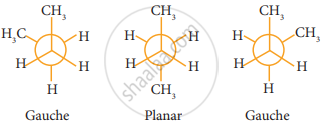
n-Butane may be considered as a derivative of ethane, as one hydrogen on each carbon is replaced by a methyl group - Eclipsed conformation:
In this conformation, the distance between the two methyl group is minimum. So there is maximum repulsion between them and it is the least stable conformer. - Anti or staggered form:
In this conformation, the distance between the two methyl groups is maximum and so there is minimum repulsion between them. And it is the most stable conformer. - The following potentially energy diagram shows the relative stabilities of various conformers of n-butane.
potential energy of a n-butane molecule as a funtion of the angle φ of bond rotation

APPEARS IN
संबंधित प्रश्न
An alkene ‘A’ on ozonolysis gives 2 moles of ethanal. Write the structure and IUPAC name of ‘A’.
Propane nitrile on reaction with ethyl magnesium iodide in presence of dry ether gives complex. This imine complex on acid hydrolysis forms ______.
Which of the following undergoes substitution reaction?
In the reaction, \[\ce{2\text{n} R - X ->[+ 2n Na][Dry ether] product}\]
The product obtained is ______.
Arrange the three isomers of alkane with molecular
formula C5H12 in increasing order of boiling points and write their IUPAC names.
How many monochlorination products are possible for 2-methylpropane?
Draw their structures and write their IUPAC names.
Write the balanced chemical reaction for the preparation of ethane from Ethyl magnesium iodide.
Arrange the three isomers of alkane with molecular formula C5 H12 in increasing order of boiling points and write their IUPAC names.
Arrange the three isomers of alkane with malecular formula \[\ce{C5H12}\] in increasing order of boiling points and write their IUPAC names.
What is the number of structural isomers for molecular formula C6H14?
