Advertisements
Advertisements
प्रश्न
Read the following reaction and answer the questions given below:

- Write the name of the reaction.
- Identify the electrophile in it.
- How is this electrophile generated?
Advertisements
उत्तर
- The name of the reaction is Friedel-Craft’s alkylation reaction.
- The electrophile in the reaction is +CH3.
- The electrophile +CH3 is generated as follows:
\[\ce{\underset{\text{Methyl chloride}}{CH3 -}Cl + AlCl3 -> \underset{\text{Electrophile}}{^+CH3 +}AlCl^-_4}\]
APPEARS IN
संबंधित प्रश्न
Predict the possible product of the following reaction.
bromination of phenol
Identify the main product of the reaction.
\[\begin{array}{cc}
\ce{CH3 - CH - CH3 ->[{60%}H2SO4][373 K]}\\
|\phantom{............}\\\ce{OH}\phantom{..........}
\end{array}\] _______.
Identify the main product of the reaction:
\[\ce{HC ≡ C - CH3 ->[H2][Pd-C/quinoline]}\] _______.
Identify giving reason whether the following compound is aromatic or not.
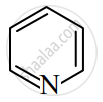
Name two reagents used for acylation of benzene.
Which of the following compounds will not undergo Friedal – crafts reaction easily ? (NEET)
Some meta-directing substituents in aromatic substitution are given. Which one is most deactivating?
2 – butyne on chlorination gives ______.
Give IUPAC name for the following compound.
Ethyl isopropyl acetylene
Give IUPAC name for the following compound.
\[\ce{CH ≡ C – C ≡ C – C ≡ CH}\]
Suggest a simple chemical test to distinguish propane and propene.
Suggest the route for the preparation of the following from benzene.
4 – chlorotoluene
What happens when Isobutylene is treated with acidified potassium permanganate?
Write the structure of the following alkanes.
5 – (1, 2 – Dimethyl propyl) – 2 – methylnonane
How will you prepare propane from a sodium salt of fatty acid?
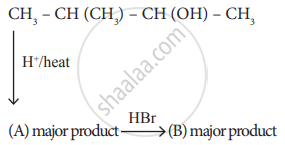
Identify A and B.
How will distinguish 1 – butyne and 2 – butyne?
When acetylene is passed through red hot iron tube at 873 K, ____________ is/are obtained as product/s.
Identify the hydrocarbon compound from following containing carbon atoms in the range of C6 to C8?
A compound with molecular formula C4H4O has all the four carbon atom and the oxygen atom in the ring. It also has two carbon-carbon double bonds. The compound is ____________.
Which among the following compounds has highest boiling point?
Phenol on distillation with zinc dust gives ____________.
According to Huckel rule, a cyclic π molecular orbital formed by overlap of p orbitals must contain ____________ p electrons.
Which of the following is NOT alicyclic compound?
Sodium benzoate on decarboxylation gives ____________.
α-butylene when subjected to hydroboration oxidation reaction, yields ____________.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..............................}\ce{CH3}\\
\phantom{...........................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide]CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{.................................}\\
\end{array}\]
- Write the IUPAC name of the product.
- State the rule that governs the formation of this product.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..............................}\ce{CH3}\\
\phantom{...........................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide] CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{.................................}
\end{array}\]
- Write the IUPAC name of the product.
- State the rule that governs the formation of this product.
