Advertisements
Advertisements
प्रश्न
Predict the possible product of the following reaction.
bromination of phenol
Advertisements
उत्तर
Phenolic –OH group is ortho and para directing group. So, the bromination of phenol gives p-bromophenol and o-bromophenol.
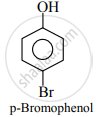
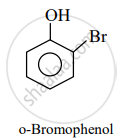
APPEARS IN
संबंधित प्रश्न
Write the balanced chemical reaction to get benzene from Sodium benzoate.
Write the balanced chemical reaction to get benzene from Phenol.
Predict the possible product of the following reaction.
sulphonation of chlorobenzene
Identify the main product of the reaction.
\[\begin{array}{cc}
\ce{CH3 - CH - CH3 ->[{60%}H2SO4][373 K]}\\
|\phantom{............}\\\ce{OH}\phantom{..........}
\end{array}\] _______.
Identify the main product of the reaction:
\[\ce{HC ≡ C - CH3 ->[H2][Pd-C/quinoline]}\] _______.
Identify giving reason whether the following compound is aromatic or not.
Identify giving reason whether the following compound is aromatic or not.
Read the following reaction and answer the questions given below:

- Write the name of the reaction.
- Identify the electrophile in it.
- How is this electrophile generated?
Which one of the following is non aromatic?
Give IUPAC name for the following compound.
\[\ce{CH ≡ C – C ≡ C – C ≡ CH}\]
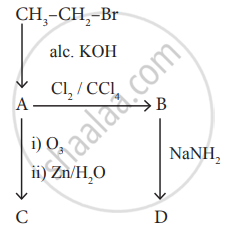
Identify the compound A, B, C and D in the following series of reactions.
How is propyne prepared from an alkylene dihalide ?
Describe the mechanism of Nitration of benzene.
Suggest a simple chemical test to distinguish propane and propene.
How does Huckel rule help to decide the aromatic character of a compound?
Suggest the route for the preparation of the following from benzene.
m - dinitro benzene
What happens when Isobutylene is treated with acidified potassium permanganate?
When acetylene is passed through red hot iron tube at 873 K, ____________ is/are obtained as product/s.
Identify the hydrocarbon compound from following containing carbon atoms in the range of C6 to C8?
−Cl group is ____________.
Phenol on distillation with zinc dust gives ____________.
Which of the following is INCORRECT for aromatic hydrocarbons?
The alkane formed on heating sodium butanoate with sodalime is ______.
α-butylene when subjected to hydroboration oxidation reaction, yields ____________.
Which of the following compounds is NOT obtained in thermal decomposition of propane in absence of air?
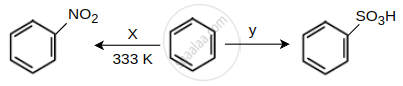
Identify the X and Y in the following reactions.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..............................}\ce{CH3}\\
\phantom{............................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide]CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{.................................}
\end{array}\]
- Write IUPAC name of the product.
- State the rule that governs formation of this product.
