Advertisements
Advertisements
प्रश्न
Write the balanced chemical reaction to get benzene from Phenol.
Advertisements
उत्तर
Phenol to benzene:
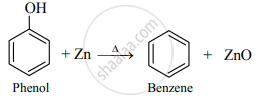
APPEARS IN
संबंधित प्रश्न
Name the following:
The hydrocarbon said to possess carcinogenic property.
Predict the possible product of the following reaction.
nitration of toluene
Identify the main product of the reaction:
\[\ce{H - C ≡ C - H + H - O ->[{40%} H2SO4][{1%} HgSO4]}\] _____.
Identify A, B, C in the following reaction sequence:
\[\ce{CH3 - CH = CH2 ->[Br2/CCl4][room temperature] A ->[Zn] B ->[dil. alkaline][KMnO4] C}\]
Major product of the below mentioned reaction is, \[\ce{(CH3 )2 C = CH2 ->[ICI]}\]
Which one of the following is non aromatic?
Which of the following compounds will not undergo Friedal – crafts reaction easily ? (NEET)
Some meta-directing substituents in aromatic substitution are given. Which one is most deactivating?
Identify the compound 'Z' in the following reaction
\[\ce{C2H6O ->[Al2O3][623 K] X ->[O3] Y ->[Zn/H2O] (Z)}\]
2 – butyne on chlorination gives ______.
Give IUPAC name for the following compound.
\[\ce{CH ≡ C – C ≡ C – C ≡ CH}\]
Write short notes on ortho, para directors in aromatic electrophilic substitution reactions.
Write the chemical equations for combustion of propane.
How does Huckel rule help to decide the aromatic character of a compound?
Suggest the route for the preparation of the following from benzene.
3 – chloro nitrobenzene
Suggest the route for the preparation of the following from benzene.
4 – chlorotoluene
Suggest the route for the preparation of the following from benzene.
Bromo benzene
What happens when Isobutylene is treated with acidified potassium permanganate?
Write the structure of the following alkanes.
5 – (1, 2 – Dimethyl propyl) – 2 – methylnonane
How will distinguish 1 – butyne and 2 – butyne?
Which of the following is NOT a hetero-aromatic compound?
Identify the hydrocarbon compound from following containing carbon atoms in the range of C6 to C8?
−Cl group is ____________.
Which of the following is NOT alicyclic compound?
Dow's process is used for the synthesis of an aromatic compound (X). Identify X.
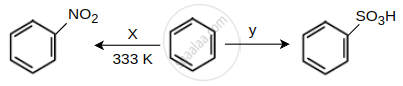
Identify the X and Y in the following reactions.
Which of the following is most stable?
