Advertisements
Advertisements
Question
Predict the possible product of the following reaction.
bromination of phenol
Advertisements
Solution
Phenolic –OH group is ortho and para directing group. So, the bromination of phenol gives p-bromophenol and o-bromophenol.
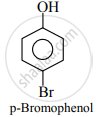
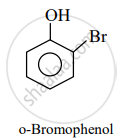
APPEARS IN
RELATED QUESTIONS
Identify the main product of the reaction:
\[\ce{H - C ≡ C - H + H - O ->[{40%} H2SO4][{1%} HgSO4]}\] _____.
Identify giving reason whether the following compound is aromatic or not.
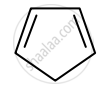
Identify giving reason whether the following compound is aromatic or not.
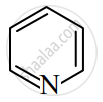
Read the following reaction and answer the questions given below:

- Write the name of the reaction.
- Identify the electrophile in it.
- How is this electrophile generated?
Identify the compound (A) in the following reaction
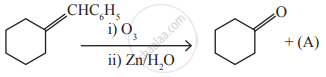
Consider the nitration of benzene using mixed con H2SO4 and HNO3 if a large quantity of KHSO4 is added to the mixture, the rate of nitration will be ______.
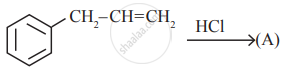 is
is
Which one of the following is non aromatic?
An alkane is obtained by decarboxylation of sodium propionate. Same alkane can be prepared by ______.
2 – butyne on chlorination gives ______.
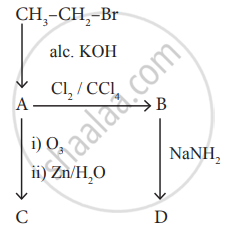
Identify the compound A, B, C and D in the following series of reactions.
How is propyne prepared from an alkylene dihalide ?
Suggest a simple chemical test to distinguish propane and propene.
Write the chemical equations for combustion of propane.
What happens when Isobutylene is treated with acidified potassium permanganate?
How will you convert ethyl chloride into n – butane?
Write the structure of the following alkanes.
5 – (1, 2 – Dimethyl propyl) – 2 – methylnonane
Complete the following:
\[\begin{array}{cc}
\ce{CH2 - CH2 ->[Zn/C2H5OH]}\\
|\phantom{.......}|\phantom{..............}\\
\ce{Br}\phantom{.....}\ce{Br}\phantom{..............}
\end{array}\]
Which of the following is NOT a hetero-aromatic compound?
The alkane formed on heating sodium butanoate with sodalime is ______.
α-butylene when subjected to hydroboration oxidation reaction, yields ____________.
Conversion of hexane into benzene involves the reaction of ______.
Which of the following statement is NOТ correct for aromatic compounds?
Identify ortho and para-directing groups from the following:
\[\ce{\underset{I}{–CHO},\underset{II}{–NHCOCH3},\underset{III}{–OCH3},\underset{IV}{–SO3H}}\]
Benzene reacts with CH3COCl + AICl3 to give ______.
