Advertisements
Advertisements
Question
Identify giving reason whether the following compound is aromatic or not.
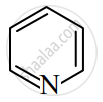
Advertisements
Solution

The compound is aromatic since it has 6π electrons and hence, obeys the Huckel rule of aromaticity.
APPEARS IN
RELATED QUESTIONS
Name the following:
The hydrocarbon said to possess carcinogenic property.
Write the balanced chemical reaction to get benzene from Phenol.
Predict the possible product of the following reaction.
sulphonation of chlorobenzene
Predict the possible product of the following reaction.
bromination of phenol
Identify giving reason whether the following compound is aromatic or not.

Identify giving reason whether the following compound is aromatic or not.
Major product of the below mentioned reaction is, \[\ce{(CH3 )2 C = CH2 ->[ICI]}\]
Consider the nitration of benzene using mixed con H2SO4 and HNO3 if a large quantity of KHSO4 is added to the mixture, the rate of nitration will be ______.
Identify the compound 'Z' in the following reaction
\[\ce{C2H6O ->[Al2O3][623 K] X ->[O3] Y ->[Zn/H2O] (Z)}\]
Write short notes on ortho, para directors in aromatic electrophilic substitution reactions.
How is propyne prepared from an alkylene dihalide ?
Describe the mechanism of Nitration of benzene.
Suggest a simple chemical test to distinguish propane and propene.
What happens when Isobutylene is treated with acidified potassium permanganate?
Write the structure of the following alkanes.
5 – (2 – Ethyl butyl) – 3, 3 – dimethyldecane
Write the structure of the following alkanes.
5 – (1, 2 – Dimethyl propyl) – 2 – methylnonane
When acetylene is passed through red hot iron tube at 873 K, ____________ is/are obtained as product/s.
Identify the hydrocarbon compound from following containing carbon atoms in the range of C6 to C8?
−Cl group is ____________.
Phenol on distillation with zinc dust gives ____________.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..............................}\ce{CH3}\\
\phantom{...........................}|\\
\ce{CH3 - C = CH2 + HBr->[Benzoyl][peroxide] H3C - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{.................................}
\end{array}\]
- Write the IUPAC name of the product.
- State the rule that governs the formation of this product.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..............................}\ce{CH3}\\
\phantom{...........................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide]CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{.................................}\\
\end{array}\]
- Write the IUPAC name of the product.
- State the rule that governs the formation of this product.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{.............................}\ce{CH3}\\
\phantom{...........................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide] CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{.................................}\\
\end{array}\]
- Write IUPAC name of the product.
- State the rule that governs formation of this product.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..............................}\ce{CH3}\\
\phantom{...........................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide] CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{.................................}
\end{array}\]
- Write the IUPAC name of the product.
- State the rule that governs the formation of this product.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{...............................}\ce{CH3}\\
\phantom{............................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide]CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{..................................}
\end{array}\]
- Write the IUPAC name of the product.
- State the rule that governs the formation of this product.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..........................}\ce{CH3}\\\phantom{........................}|\\\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide] CH3 - CH - CH2Br}\\
|\phantom{......................................}\\
\ce{CH3}\phantom{....................................}\end{array}\]
- Write the IUPAC name of the product.
- State the rule that governs the formation of this product.
Identify ortho and para-directing groups from the following:
\[\ce{\underset{I}{–CHO},\underset{II}{–NHCOCH3},\underset{III}{–OCH3},\underset{IV}{–SO3H}}\]
