Advertisements
Advertisements
प्रश्न
How will you convert ethyl chloride into ethane?
Advertisements
उत्तर
\[\ce{\underset{\text{ethyl chloride}}{C2H5Cl} ->[Zn/HCl][(H)] C2H6 \underset{\text{ethane}}{+ HCl}}\]
APPEARS IN
संबंधित प्रश्न
Name two reagents used for acylation of benzene.
Which of the following compounds will not undergo Friedal – crafts reaction easily ? (NEET)
Which of the following can be used as the halide component for friedal - crafts reaction?
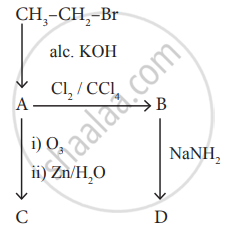
Identify the compound A, B, C and D in the following series of reactions.
Describe the mechanism of Nitration of benzene.
How will distinguish 1 – butyne and 2 – butyne?
A compound with molecular formula C4H4O has all the four carbon atom and the oxygen atom in the ring. It also has two carbon-carbon double bonds. The compound is ____________.
−Cl group is ____________.
Phenol on distillation with zinc dust gives ____________.
According to Huckel rule, a cyclic π molecular orbital formed by overlap of p orbitals must contain ____________ p electrons.
