Advertisements
Advertisements
Question
Identify the main product of the reaction.
\[\begin{array}{cc}
\ce{CH3 - CH - CH3 ->[{60%}H2SO4][373 K]}\\
|\phantom{............}\\\ce{OH}\phantom{..........}
\end{array}\] _______.
Advertisements
Solution
\[\begin{array}{cc}
\ce{CH3 - CH - CH3 ->[{60%}H2SO4][373 K] CH3 \underset{\text{Propene}}{- CH =} CH2 + H2O}\\
|\phantom{......................................}\\\ce{\underset{\text{Propan-2-ol}}{OH}}\phantom{....................................}
\end{array}\]
APPEARS IN
RELATED QUESTIONS
Name the following:
The hydrocarbon said to possess carcinogenic property.
Write the balanced chemical reaction to get benzene from Sodium benzoate.
Write the balanced chemical reaction to get benzene from Phenol.
Predict the possible product of the following reaction.
Chlorination of nitrobenzene
Predict the possible product of the following reaction.
nitration of toluene
Identify giving reason whether the following compound is aromatic or not.
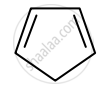
Identify giving reason whether the following compound is aromatic or not.
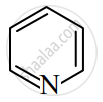
Name two reagents used for acylation of benzene.
Which of the following compounds will not undergo Friedal – crafts reaction easily ? (NEET)
Which of the following can be used as the halide component for friedal - crafts reaction?
2 – butyne on chlorination gives ______.
Write short notes on ortho, para directors in aromatic electrophilic substitution reactions.
Write the chemical equations for combustion of propane.
What happens when Isobutylene is treated with acidified potassium permanganate?
How will you convert ethyl chloride into ethane?
Write the structure of the following alkanes.
5 – (2 – Ethyl butyl) – 3, 3 – dimethyldecane
Identify the hydrocarbon compound from following containing carbon atoms in the range of C6 to C8?
Which of the following is INCORRECT for aromatic hydrocarbons?
Which of the following is NOT alicyclic compound?
Which of the following compounds on bromination yields ![]() ?
?
The treatment of benzene with iso - butene in the presence of sulphuric acid gives:
Conversion of hexane into benzene involves the reaction of ______.
Arenes on treatments with chlorine in the presence of ferric chloride as a catalyst undergo what type of reaction?
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..............................}\ce{CH3}\\
\phantom{...........................}|\\
\ce{CH3 - C = CH2 + HBr ->[Benzoyl][peroxide] H3C - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{.................................}
\end{array}\]
- Write the IUPAC name of the product.
- State the rule that governs the formation of this product.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{.............................}\ce{CH3}\\
\phantom{...........................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide] CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{.................................}\\
\end{array}\]
- Write IUPAC name of the product.
- State the rule that governs formation of this product.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..............................}\ce{CH3}\\
\phantom{............................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide] CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH_3}\phantom{.................................}
\end{array}\]
- Write the IUPAC name of the product.
- State the rule that governs the formation of this product.
Identify ortho and para-directing groups from the following:
\[\ce{\underset{I}{–CHO},\underset{II}{–NHCOCH3},\underset{III}{–OCH3},\underset{IV}{–SO3H}}\]
