Advertisements
Advertisements
Question
Complete the following:
\[\begin{array}{cc}
\ce{CH2 - CH2 ->[Zn/C2H5OH]}\\
|\phantom{.......}|\phantom{..............}\\
\ce{Br}\phantom{.....}\ce{Br}\phantom{..............}
\end{array}\]
Advertisements
Solution
\[\begin{array}{cc}
\ce{CH2 - CH2 ->[Zn/C2H5OH] CH2 \underset{\text{Ethylene}}{= CH2 + ZnBr2}}\\
|\phantom{.......}|\phantom{..................................}\\
\ce{Br}\phantom{.....}\ce{Br}\phantom{.................................}
\end{array}\]
APPEARS IN
RELATED QUESTIONS
Write the balanced chemical reaction to get benzene from Sodium benzoate.
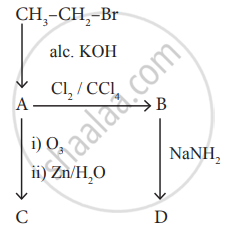
Identify the compound A, B, C and D in the following series of reactions.
How will you convert ethyl chloride into ethane?
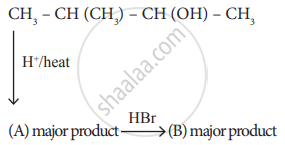
Identify A and B.
A compound with molecular formula C4H4O has all the four carbon atom and the oxygen atom in the ring. It also has two carbon-carbon double bonds. The compound is ____________.
Sodium benzoate on decarboxylation gives ____________.
Which of the following compounds is NOT obtained in thermal decomposition of propane in absence of air?
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..............................}\ce{CH3}\\
\phantom{...........................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide] CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{.................................}
\end{array}\]
- Write the IUPAC name of the product.
- State the rule that governs the formation of this product.
Which of the following statement is NOТ correct for aromatic compounds?
Identify 'A' in the following reaction.
\[\ce{\underset{}{A + Chloromethane} ->[anhydrous][AlCl3] \underset{\underset{}{}}{2-Chlorotoluene + 4-Chlorotoluene}}\]
