Advertisements
Advertisements
Question
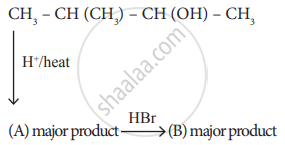
Identify A and B.
Advertisements
Solution
A)
\[\begin{array}{cc}
\ce{CH3 - C = CH - CH3 (2 - methyl 2 - bytene)}\\|\phantom{..............................}\\
\ce{CH3}\phantom{............................}
\end{array}\]
B)
\[\begin{array}{cc}\phantom{.........}\ce{CH3}\\
\phantom{.......}|\\\ce{CH3 - CH2 - C - CH3}\\
\phantom{.......}|\\\phantom{........}\ce{Br}
\end{array}\]
2 - bromo 2 methyl butane
APPEARS IN
RELATED QUESTIONS
Predict the possible product of the following reaction.
nitration of toluene
Identify the main product of the reaction.
\[\begin{array}{cc}
\ce{CH3 - CH - CH3 ->[{60%}H2SO4][373 K]}\\
|\phantom{............}\\\ce{OH}\phantom{..........}
\end{array}\] _______.
Consider the nitration of benzene using mixed con H2SO4 and HNO3 if a large quantity of KHSO4 is added to the mixture, the rate of nitration will be ______.
Describe the mechanism of Nitration of benzene.
Write the chemical equations for combustion of propane.
How does Huckel rule help to decide the aromatic character of a compound?
How will you convert ethyl chloride into n – butane?
Complete the following:
\[\begin{array}{cc}
\ce{CH2 - CH2 ->[Zn/C2H5OH]}\\
|\phantom{.......}|\phantom{..............}\\
\ce{Br}\phantom{.....}\ce{Br}\phantom{..............}
\end{array}\]
Which of the following is NOT alicyclic compound?
Which of the following statement is NOТ correct for aromatic compounds?
