Advertisements
Advertisements
Question
How will you prepare propane from a sodium salt of fatty acid?
Advertisements
Solution
\[\ce{CH3 \underset{\text{(Sodium Salt of butyric acid)}}{- CH2 - CH2 - COONa} ->[NaOH/CaO] \underset{\text{(Propane)}}{CH3 - CH2} - CH3 + Na2CO3}\]
Sodium salt of butyric acid on heating with sodalime gives propane.
APPEARS IN
RELATED QUESTIONS
Identify giving reason whether the following compound is aromatic or not.
Major product of the below mentioned reaction is, \[\ce{(CH3 )2 C = CH2 ->[ICI]}\]
Which of the following can be used as the halide component for friedal - crafts reaction?
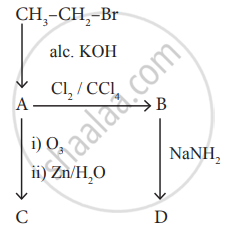
Identify the compound A, B, C and D in the following series of reactions.
Suggest the route for the preparation of the following from benzene.
3 – chloro nitrobenzene
Suggest the route for the preparation of the following from benzene.
Bromo benzene
What happens when Isobutylene is treated with acidified potassium permanganate?
According to Huckel rule, a cyclic π molecular orbital formed by overlap of p orbitals must contain ____________ p electrons.
Which of the following is INCORRECT for aromatic hydrocarbons?
Which of the following molecules has shortest C - C bond length?
