Advertisements
Advertisements
प्रश्न
How will you prepare propane from a sodium salt of fatty acid?
Advertisements
उत्तर
\[\ce{CH3 \underset{\text{(Sodium Salt of butyric acid)}}{- CH2 - CH2 - COONa} ->[NaOH/CaO] \underset{\text{(Propane)}}{CH3 - CH2} - CH3 + Na2CO3}\]
Sodium salt of butyric acid on heating with sodalime gives propane.
APPEARS IN
संबंधित प्रश्न
Identify the main product of the reaction:
\[\ce{H - C ≡ C - H + H - O ->[{40%} H2SO4][{1%} HgSO4]}\] _____.
Identify giving reason whether the following compound is aromatic or not.
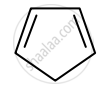
Identify giving reason whether the following compound is aromatic or not.

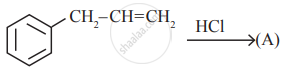 is
is
Which of the following is aliphatic saturated hydrocarbon?
Give IUPAC name for the following compound.
Ethyl isopropyl acetylene
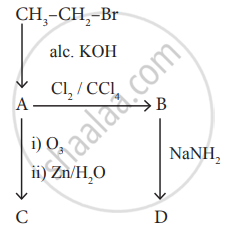
Identify the compound A, B, C and D in the following series of reactions.
Write the structure of the following alkanes.
2, 3 – Dimethyl – 6 – (2 – methyl propyl) decane
When acetylene is passed through red hot iron tube at 873 K, ____________ is/are obtained as product/s.
Which of the following compounds on bromination yields ![]() ?
?
