Advertisements
Advertisements
प्रश्न
Name two reagents used for acylation of benzene.
Advertisements
उत्तर
The two reagents used for acylation of benzene are:
- CH3COCl (acetyl chloride) and anhydrous AlCl3
- (CH3CO)2O (acetic anhydride) and anhydrous AlCl3
APPEARS IN
संबंधित प्रश्न
Predict the possible product of the following reaction.
sulphonation of chlorobenzene
Read the following reaction and answer the questions given below:

- Write the name of the reaction.
- Identify the electrophile in it.
- How is this electrophile generated?
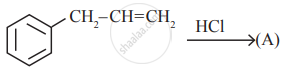 is
is
2 – butyne on chlorination gives ______.
Give IUPAC name for the following compound.
Ethyl isopropyl acetylene
Give IUPAC name for the following compound.
\[\ce{CH ≡ C – C ≡ C – C ≡ CH}\]
Write short notes on ortho, para directors in aromatic electrophilic substitution reactions.
How will you convert ethyl chloride into n – butane?
Complete the following:
\[\begin{array}{cc}
\ce{CH2 - CH2 ->[Zn/C2H5OH]}\\
|\phantom{.......}|\phantom{..............}\\
\ce{Br}\phantom{.....}\ce{Br}\phantom{..............}
\end{array}\]
Which of the following is an aromatic compound?
A compound with molecular formula C4H4O has all the four carbon atom and the oxygen atom in the ring. It also has two carbon-carbon double bonds. The compound is ____________.
Which among the following compounds has highest boiling point?
−Cl group is ____________.
Which of the following is NOT alicyclic compound?
The alkane formed on heating sodium butanoate with sodalime is ______.
Dow's process is used for the synthesis of an aromatic compound (X). Identify X.
Which of the following compounds on bromination yields ![]() ?
?
Arenes on treatments with chlorine in the presence of ferric chloride as a catalyst undergo what type of reaction?
Identify the X and Y in the following reactions.
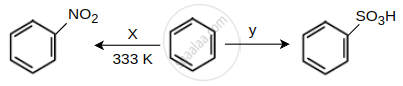
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{...............................}\ce{CH3}\\
\phantom{............................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide]CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{..................................}
\end{array}\]
- Write the IUPAC name of the product.
- State the rule that governs the formation of this product.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..........................}\ce{CH3}\\\phantom{........................}|\\\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide] CH3 - CH - CH2Br}\\
|\phantom{......................................}\\
\ce{CH3}\phantom{....................................}\end{array}\]
- Write the IUPAC name of the product.
- State the rule that governs the formation of this product.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..............................}\ce{CH3}\\
\phantom{............................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide] CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{..................................}
\end{array}\]
- Write IUPAC name of the product.
- State the rule that governs formation of this product.
Identify 'A' in the following reaction.
\[\ce{\underset{}{A + Chloromethane} ->[anhydrous][AlCl3] \underset{\underset{}{}}{2-Chlorotoluene + 4-Chlorotoluene}}\]
Identify ortho and para-directing groups from the following:
\[\ce{\underset{I}{–CHO},\underset{II}{–NHCOCH3},\underset{III}{–OCH3},\underset{IV}{–SO3H}}\]
