Advertisements
Advertisements
प्रश्न
Identify giving reason whether the following compound is aromatic or not.
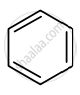
Advertisements
उत्तर
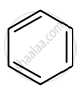
The compound is aromatic since it has 6π electrons and hence, obeys the Huckel rule of aromaticity.
APPEARS IN
संबंधित प्रश्न
Name the following:
The hydrocarbon said to possess carcinogenic property.
Write the balanced chemical reaction to get benzene from Phenol.
Identify the main product of the reaction.
\[\begin{array}{cc}
\ce{CH3 - CH - CH3 ->[{60%}H2SO4][373 K]}\\
|\phantom{............}\\\ce{OH}\phantom{..........}
\end{array}\] _______.
Identify the main product of the reaction:
\[\ce{H - C ≡ C - H + H - O ->[{40%} H2SO4][{1%} HgSO4]}\] _____.
Name two reagents used for acylation of benzene.
Which one of the following is non aromatic?
Identify the compound 'Z' in the following reaction
\[\ce{C2H6O ->[Al2O3][623 K] X ->[O3] Y ->[Zn/H2O] (Z)}\]
Write short notes on ortho, para directors in aromatic electrophilic substitution reactions.
Describe the mechanism of Nitration of benzene.
Suggest the route for the preparation of the following from benzene.
m - dinitro benzene
What happens when Isobutylene is treated with acidified potassium permanganate?
Write the structure of the following alkanes.
5 – (1, 2 – Dimethyl propyl) – 2 – methylnonane
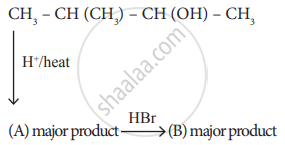
Identify A and B.
Which of the following is an aromatic compound?
Phenol on distillation with zinc dust gives ____________.
According to Huckel rule, a cyclic π molecular orbital formed by overlap of p orbitals must contain ____________ p electrons.
Sodium benzoate on decarboxylation gives ____________.
α-butylene when subjected to hydroboration oxidation reaction, yields ____________.
Which of the following molecules has shortest C - C bond length?
Which of the following reagents can bring about following conversion?
\[\ce{But-1-ene -> Butan-2-ol}\]
Which of the following compounds on bromination yields ![]() ?
?
Conversion of hexane into benzene involves the reaction of ______.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{.............................}\ce{CH3}\\
\phantom{...........................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide] CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{.................................}\\
\end{array}\]
- Write IUPAC name of the product.
- State the rule that governs formation of this product.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..............................}\ce{CH3}\\
\phantom{............................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide] CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH_3}\phantom{.................................}
\end{array}\]
- Write the IUPAC name of the product.
- State the rule that governs the formation of this product.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..............................}\ce{CH3}\\
\phantom{...........................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide] CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{.................................}
\end{array}\]
- Write the IUPAC name of the product.
- State the rule that governs the formation of this product.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..............................}\ce{CH3}\\
\phantom{............................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide] CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{..................................}
\end{array}\]
- Write IUPAC name of the product.
- State the rule that governs formation of this product.
Which of the following statement is NOТ correct for aromatic compounds?
Identify 'A' in the following reaction.
\[\ce{\underset{}{A + Chloromethane} ->[anhydrous][AlCl3] \underset{\underset{}{}}{2-Chlorotoluene + 4-Chlorotoluene}}\]
