Advertisements
Advertisements
प्रश्न
Write the chemical equations for combustion of propane.
Advertisements
उत्तर
\[\ce{\underset{\text{propane}}{C3H8(g)} + 5O2(g) ) → 3CO2(g) + 4H2O(l)}\]
APPEARS IN
संबंधित प्रश्न
Write the balanced chemical reaction to get benzene from Phenol.
Predict the possible product of the following reaction.
bromination of phenol
Identify giving reason whether the following compound is aromatic or not.
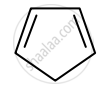
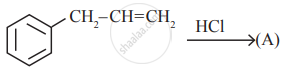 is
is
How will you convert ethyl chloride into ethane?
Complete the following:
\[\begin{array}{cc}
\ce{CH2 - CH2 ->[Zn/C2H5OH]}\\
|\phantom{.......}|\phantom{..............}\\
\ce{Br}\phantom{.....}\ce{Br}\phantom{..............}
\end{array}\]
Which of the following is NOT alicyclic compound?
Sodium benzoate on decarboxylation gives ____________.
Which of the following compounds on bromination yields ![]() ?
?
Conversion of hexane into benzene involves the reaction of ______.
