Advertisements
Advertisements
Question
Differentiate between the reversible and irreversible processes.
Advertisements
Solution
| Sr. No. | Reversible process | Irreversible process |
| i. | A reversible process is a change that can be retraced in a reverse (opposite) direction. | An irreversible process is a change that cannot be retraced in a reverse (opposite) direction. |
| ii. | The path of a reversible process is the same in the forward and the reverse direction. | The path of an irreversible process is not the same in the forward and the reverse direction. |
| iii. | Reversible changes are very slow and there is no loss of any energy in the process. | There is a permanent loss of energy from the system due to friction or other dissipative forces in an irreversible process. |
| iv. | The system comes back to its initial state after it is taken along the reverse path. | The change of state depends upon the path taken to change the state during an irreversible process. |
| v. | Reversible processes are ideal processes. | Irreversible processes are real processes. |
| vi. |
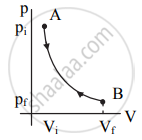
p-V diagram:
|
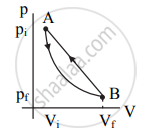
p-V diagram:
|
RELATED QUESTIONS
Explain why The climate of a harbour town is more temperate than that of a town in a desert at the same latitude.
A thermodynamic system is taken from an original state to an intermediate state by the linear process shown in Figure

Its volume is then reduced to the original value from E to F by an isobaric process. Calculate the total work done by the gas from D to E to F
An ideal gas is taken through an isothermal process. If it does 2000 J of work on its environment, how much heat is added to it?
For work done to be reversible, the process should be ______
What is a thermodynamic process?
Draw a p-V diagram of the reversible process.
Draw a p-V diagram showing positive work with varying pressure.
Explain graphically (i) positive work with varying pressure, (ii) negative work with varying pressure, and (iii) positive work at constant pressure.
When a cycle tyre suddenly bursts, the air inside the tyre expands. This process is ____________.
When you exercise in the morning, by considering your body as a thermodynamic system, which of the following is true?
The V-T diagram of an ideal gas which goes through a reversible cycle A→B→C→D is shown below. (Processes D→A and B→C are adiabatic)

The corresponding PV diagram for the process is (all figures are schematic)
Apply first law for an isothermal process.
Apply first law for an isobaric process.
Give the equation of state for an adiabatic process.
Give an equation state for an isochoric process.
Draw the PV diagram for the isochoric process.
What is a cyclic process?
Explain in detail the isothermal process.
Derive the work done in an adiabatic process.
Explain the isobaric process and derive the work done in this process.
Explain in detail the isochoric process.
Draw the TP diagram (P-x axis, T-y axis), VT(T-x axis, V-y axis) diagram for
- Isochoric process
- Isothermal process
- Isobaric process
Consider the following cyclic process consist of isotherm, isochoric and isobar which is given in the figure.

Draw the same cyclic process qualitatively in the V-T diagram where T is taken along the x-direction and V is taken along the y-direction. Analyze the nature of heat exchange in each process.
For a given ideal gas 6 × 105 J heat energy is supplied and the volume of gas is increased from 4 m3 to 6 m3 at atmospheric pressure. Calculate
- the work done by the gas
- change in internal energy of the gas
- graph this process in PV and TV diagram
An ideal gas is expanded isothermally from volume V1 to volume V2 and then compressed adiabatically to original volume V1. If the initial pressure is P1, the final pressure is P3 and net work done is W, then ____________.
In which of the following processes, beat is neither absorbed nor released by a system?
An ideal gas A and a real gas B have their volumes increased from V to 2V under isothermal conditions. The increase in internal energy ____________.
An ideal gas is compressed to half its initial volume by means of several processes. Which of the process results in the maximum work done on the gas?
Ideal gas for which 'ϒ' = 1.5 is suddenly compressed to `1/4`th of its initial volume. The ratio of 4 the final pressure to the initial pressure is ______.
`(ϒ = "C"_"p"/"C"_"v")`