Advertisements
Advertisements
Question
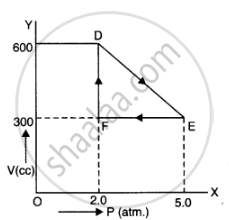
A thermodynamic system is taken from an original state to an intermediate state by the linear process shown in Figure

Its volume is then reduced to the original value from E to F by an isobaric process. Calculate the total work done by the gas from D to E to F
Advertisements
Solution 1
Total work done by the gas from D to E to F = Area of ΔDEF
Area of ΔDEF =`1/2DExxEF`
Where,
DF = Change in pressure
= 600 N/m2 – 300 N/m2
= 300 N/m2
FE = Change in volume
= 5.0 m3 – 2.0 m3
= 3.0 m3
Area of ΔDEF =`1/2xx300xx3` = 450 J
Therefore, the total work done by the gas from D to E to F is 450 J.
Solution 2
As is clear figure
Change in pressure, `triangle P = EF = 5.0 - 2.0 = 3.0 atm = 3.0 xx 10^5 Nm^(-2)`
Change in volume. triangle V = DF = 600 - 300 = 300 cc
= `300 xx 10^(-6) m^3`
Work done by the gas from D to E to F = area of `triangle DEF`
`W =1/2 xx DF xx EF`
`= 1/2 xx (300 xx 10^(-6)) xx (3.0 xx 10^5) = 45 J`
RELATED QUESTIONS
For work done to be reversible, the process should be ______
Draw a p-V diagram showing positive work at constant pressure.
3 mole of a gas at temperature 400 K expands isothermally from an initial volume of 4 litres to a final volume of 8 litres. Find the work done by the gas. (R = 8.31 J mol-1 K-1)
An ideal gas of volume 2 L is adiabatically compressed to (1/10)th of its initial volume. Its initial pressure is 1.01 x 105 Pa, calculate the final pressure. (Given 𝛾 = 1.4)
When food is cooked in a vessel by keeping the lid closed, after some time the steam pushes the lid outward. By considering the steam as a thermodynamic system, then in the cooking process
The V-T diagram of an ideal gas which goes through a reversible cycle A→B→C→D is shown below. (Processes D→A and B→C are adiabatic)

The corresponding PV diagram for the process is (all figures are schematic)
Give the equation of state for an isothermal process.
Apply first law for an isothermal process.
Give the equation of state for an adiabatic process.
Give an equation state for an isochoric process.
What is meant by a reversible and irreversible processes?
Explain in detail the isothermal process.
Derive the work done in an adiabatic process.
In an adiabatic expansion of the air, the volume is increased by 4%, what is the percentage change in pressure? (For air γ = 1.4)
In a petrol engine, (internal combustion engine) air at atmospheric pressure and temperature of 20°C is compressed in the cylinder by the piston to `1/8` of its original volume. Calculate the temperature of the compressed air. (For air γ = 1.4)
Among the amount of heat absorbed and the amount of work done by a system, ______
For an isothermal expansion of a perfect gas, the value of `(Delta "P")/"P"` is equal to ____________.
