Advertisements
Advertisements
Question
When food is cooked in a vessel by keeping the lid closed, after some time the steam pushes the lid outward. By considering the steam as a thermodynamic system, then in the cooking process
Options
Q > 0, W > 0
Q < 0, W > 0
Q > 0, W < 0
Q < 0, W < 0
Advertisements
Solution
Q > 0, W > 0
APPEARS IN
RELATED QUESTIONS
Give an example of some familiar process in which heat is added to an object, without changing its temperature.
Explain the cyclic process.
An ideal gas of volume 2 L is adiabatically compressed to (1/10)th of its initial volume. Its initial pressure is 1.01 x 105 Pa, calculate the final pressure. (Given 𝛾 = 1.4)
Explain thermodynamics of the adiabatic process.
In an isochoric process, we have ____________.
What are the limitations of the first law of thermodynamics?
Consider the following cyclic process consist of isotherm, isochoric and isobar which is given in the figure.

Draw the same cyclic process qualitatively in the V-T diagram where T is taken along the x-direction and V is taken along the y-direction. Analyze the nature of heat exchange in each process.
Ideal gas for which 'ϒ' = 1.5 is suddenly compressed to `1/4`th of its initial volume. The ratio of 4 the final pressure to the initial pressure is ______.
`(ϒ = "C"_"p"/"C"_"v")`
The work done on the system in changing the state of a gas adiabatically from equilibrium state A to equilibrium state B is 22.4 J. If the gas is taken from state A to B through another process in which the net heat absorbed by the system is 15.5 cal, then the net work done by the system in the latter case is ______.
( l cal = 4.2 J)
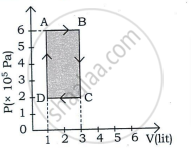
An ideal gas is taken through a cyclic process ABCDA as shown in figure. The net work done by the gas during the cycle is ______.