Advertisements
Advertisements
प्रश्न
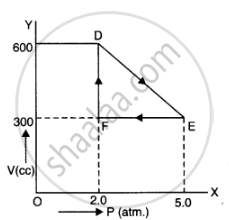
A thermodynamic system is taken from an original state to an intermediate state by the linear process shown in Figure

Its volume is then reduced to the original value from E to F by an isobaric process. Calculate the total work done by the gas from D to E to F
Advertisements
उत्तर १
Total work done by the gas from D to E to F = Area of ΔDEF
Area of ΔDEF =`1/2DExxEF`
Where,
DF = Change in pressure
= 600 N/m2 – 300 N/m2
= 300 N/m2
FE = Change in volume
= 5.0 m3 – 2.0 m3
= 3.0 m3
Area of ΔDEF =`1/2xx300xx3` = 450 J
Therefore, the total work done by the gas from D to E to F is 450 J.
उत्तर २
As is clear figure
Change in pressure, `triangle P = EF = 5.0 - 2.0 = 3.0 atm = 3.0 xx 10^5 Nm^(-2)`
Change in volume. triangle V = DF = 600 - 300 = 300 cc
= `300 xx 10^(-6) m^3`
Work done by the gas from D to E to F = area of `triangle DEF`
`W =1/2 xx DF xx EF`
`= 1/2 xx (300 xx 10^(-6)) xx (3.0 xx 10^5) = 45 J`
संबंधित प्रश्न
Explain why The climate of a harbour town is more temperate than that of a town in a desert at the same latitude.
For work done to be reversible, the process should be ______
Heating a gas in a constant volume container is an example of which process?
What is a thermodynamic process?
Draw a p-V diagram showing positive work with varying pressure.
State the assumptions made for thermodynamic processes.
An ideal gas of volume 2 L is adiabatically compressed to (1/10)th of its initial volume. Its initial pressure is 1.01 x 105 Pa, calculate the final pressure. (Given 𝛾 = 1.4)
Explain thermodynamics of the adiabatic process.
Apply first law for an adiabatic process.
Give an equation state for an isochoric process.
Explain in detail the isothermal process.
In an adiabatic expansion of the air, the volume is increased by 4%, what is the percentage change in pressure? (For air γ = 1.4)
A thermodynamic system undergoes cyclic process ABCDA as shown in the figure. The work done by the system is ______

A monoatomic gas of pressure p having volume V expands isothermally to a volume 2V and then adiabatically to a volume 16V. The final pressure of the gas is ____________.
`("ratio of specific heats" = 5/3)`
Among the amount of heat absorbed and the amount of work done by a system, ______
Two identical samples of a gas are allowed to expand (i) isothermally (ii) adiabatically. Work done is ____________.
An ideal gas A and a real gas B have their volumes increased from V to 2V under isothermal conditions. The increase in internal energy ____________.
An ideal gas is compressed to half its initial volume by means of several processes. Which of the process results in the maximum work done on the gas?
