Advertisements
Advertisements
Question
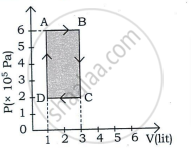
Draw the PV diagram for the isochoric process.
Diagram
Advertisements
Solution

shaalaa.com
Is there an error in this question or solution?
Chapter 8: Heat and Thermodynamics - Evaluation [Page 158]
APPEARS IN
RELATED QUESTIONS
Draw a p-V diagram showing positive work with varying pressure.
Draw a p-V diagram showing positive work at constant pressure.
Explain the thermodynamics of the isochoric process.
Explain thermodynamics of the adiabatic process.
Apply first law for an isothermal process.
Give an equation state for an isochoric process.
Derive the work done in an adiabatic process.
Two identical samples of a gas are allowed to expand (i) isothermally (ii) adiabatically. Work done is ____________.
In which of the following processes, beat is neither absorbed nor released by a system?
An ideal gas is taken through a cyclic process ABCDA as shown in figure. The net work done by the gas during the cycle is ______.