Advertisements
Advertisements
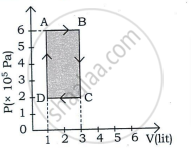
प्रश्न
Draw the PV diagram for the isochoric process.
Advertisements
उत्तर

APPEARS IN
संबंधित प्रश्न
A thermodynamic system is taken from an original state to an intermediate state by the linear process shown in Figure

Its volume is then reduced to the original value from E to F by an isobaric process. Calculate the total work done by the gas from D to E to F
Give the equation of state for an adiabatic process.
Draw the PV diagram for the isothermal process.
What is a cyclic process?
Explain in detail the isothermal process.
A thermodynamic system undergoes cyclic process ABCDA as shown in the figure. The work done by the system is ______

Two identical samples of a gas are allowed to expand (i) isothermally (ii) adiabatically. Work done is ____________.
Which of the following processes is reversible?
An ideal gas is taken through a cyclic process ABCDA as shown in figure. The net work done by the gas during the cycle is ______.
In a cyclic process, if ΔU = internal energy, W = work done, Q = Heat supplied then ______.
