Topics
Gravitation
- Concept of Gravitation
- Force and Motion
- Centripetal Force
- Kepler’s Laws
- Law of Orbit or Kepler's First Law
- Law of Areas or Kepler's Second Law
- Law of Periods or Kepler's Third Law
- Newton's Universal Law of Gravitation
- Uniform Circular Motion (UCM)
- Earth’s Gravitational Force
- Earth’s Gravitational Acceleration
- Mass and Weight
- Gravitational Waves
- Free Fall
- Gravitational Potential Energy
- Escape Velocity
- Weightlessness in Space
Periodic Classification of Elements
- Classification of Elements
- Dobereiner’s Triads
- Newland's Law of Octaves
- Mendeleev’s Periodic Table
- Insights into Mendeleev’s Periodic Table
- Modern Periodic Law
- The Modern Periodic Table
- Structure of the Modern Periodic Table
- Modern Periodic Table and Electronic Configuration of Elements
- Groups and Electronic Configuration
- Periods and Electronic Configuration
- Periodic Trends in the Modern Periodic Table
- Atomic Size
- Metallic and Non-metallic Characters
- Gradation in Halogen Family
Chemical Reactions and Equations
- Chemical Reaction
- Chemical Equations
- Balancing Chemical Equation
- Types of Chemical Reactions > Combination Reaction
- Types of Chemical Reactions > Decomposition Reaction
- Types of Chemical Reactions > Single Displacement Reaction
- Types of Chemical Reactions > Double Displacement Reaction
- Endothermic and Exothermic Processes
- Rate of Chemical Reaction
- Factors Affecting the Rate of a Chemical Reaction
- Chemical Properties of Carbon Compounds > Oxidation
- Chemical Properties of Carbon Compounds > Reduction
- Corrosion of Metals
- Rancidity
Effects of Electric Current
- Electric Circuit
- Heating Effect of Electric Current
- Magnetic Effect of Electric Current
- Right-hand Thumb Rule
- Applications of Biot-Savart's Law > Magnetic Field at the Centre of a Circular Loop
- Applications of Ampere’s Circuital Law > Magnetic Field of a Long Straight Solenoid
- Force on a Current Carrying Conductor in a Magnetic Field
- Fleming’s Left Hand Rule
- Electric Motor
- Electromagnetic Induction
- Galvanometer
- Faraday's Laws of Electromagnetic Induction
- Fleming’s Right Hand Rule
- Alternating current (AC) and Direct Current (DC)
- Electric Generator
Heat
Refraction of Light
Lenses
- Concept of Lenses
- Images Formed by Convex Lenses
- Images Formed by Concave Lenses
- Sign Convention
- Lens Formula
- Magnification
- Power of a Lens
- Combination of Lenses
- Defects of Vision and Their Corrections > Myopia
- Defects of Vision and Their Corrections > Hypermetropia
- Defects of Vision and Their Corrections > Presbyopia
- Apparent Size of an Object
- Use of Concave Lenses
- Use of Convex Lenses
- Persistence of Vision
Metallurgy
- Physical Properties of Metals
- Physical Properties of Non-metal
- Chemical Properties of Metal
- Reactions of Metals
- Reactivity Series of Metals
- Chemical Properties of Non-metal
- Ionic Compounds
- Metallurgy
- Basic Principles of Metallurgy > Concentration of Ores
- Basic Principles of Metallurgy > Extraction of Metals
- Basic Principles of Metallurgy > Refining of Metals
- Corrosion of Metals
- Prevention of Corrosion
Carbon Compounds
- Bonds in Carbon Compounds
- Carbon: A Versatile Element
- Hydrocarbons
- Straight chains, Branched chains, and Rings of Carbon atoms
- Functional Groups in Carbon Compounds
- Homologous Series
- Nomenclature
- Chemical Properties of Carbon Compounds > Combustion
- Chemical Properties of Carbon Compounds > Oxidation
- Chemical Properties of Carbon Compounds > Addition Reaction
- Chemical Properties of Carbon Compounds > Substitution Reaction
- Ethanol
- Ethanoic Acid
- Macromolecules and Polymers
Space Missions
School of Elements
The Magic of Chemical Reactions
- Chemical Equations
- Types of Chemical Reactions > Combination Reaction
- Types of Chemical Reactions > Decomposition Reaction
- Types of Chemical Reactions > Single Displacement Reaction
- Types of Chemical Reactions > Double Displacement Reaction
- Chemical Properties of Carbon Compounds > Oxidation
- Types of Double Displacement: Neutralization Reaction
The Acid Base Chemistry
- Properties of Acids > Physical Properties
- The pH Scale
- Acids, Bases and Their Reactivity
- Acid or a Base in a Water Solution
- Preparation and Uses of Baking Soda
- Preparation and Uses of Bleaching Powder
- Preparation and Uses of Washing Soda
- Preparation and Uses of Plaster of Paris
- Chemicals from Common Salt - Soap as a Salt
The Electric Spark
All about Electromagnetism
- Magnetic force
- Bar Magnet and Solenoid Analogy
- Right-hand Thumb Rule
- Applications of Biot-Savart's Law > Magnetic Field at the Centre of a Circular Loop
- Applications of Ampere’s Circuital Law > Magnetic Field of a Long Straight Solenoid
- Force on a Current Carrying Conductor in a Magnetic Field
- Electric Motor
- Electromagnetic Induction
- A.C. Generator
- Simple D.C. Motor
- Household Electrical Circuits
Wonders of Light 1
Wonders of Light 2
Striving for better Environment 1
- Abatement of Pollution
- Sustainable Use of Resources
- Definition: Organic Compounds
- Definition: Covalent Bond
- Key Points: Bonds in Carbon Compounds
Properties and Forms of Carbon
Carbon is a fundamental element present in all living organisms and many non-living substances. Organic compounds contain carbon, except for a few inorganic carbon compounds like carbonates, bicarbonates, and oxides of carbon.
- Atomic number: 6
- Atomic mass: 12.01 g/mol
- Found in both free (coal, graphite) and combined states (carbonates, hydrocarbons, CO₂).
- Forms bonds with elements like hydrogen, oxygen, chlorine, and sulphur, creating various compounds.
Catenation Property of Carbon:
Carbon has a unique ability to form long chains and rings by bonding with itself. This property is called catenation. Additionally, carbon can form double and triple bonds with elements like oxygen and nitrogen, leading to diverse organic compounds.

Allotropes of Carbon:
Carbon exists in multiple forms called allotropes, which have different physical properties but similar chemical behaviour.
|
Diamond: the hardest known material, used in jewellery and cutting tools. |
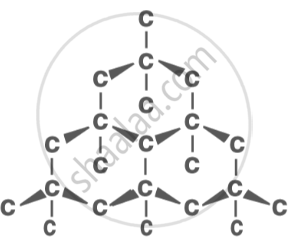 |
|
Graphite: a good conductor of electricity, used in pencils and lubricants. |
 |
|
Graphene: a single-layer structure with high electrical conductivity. |
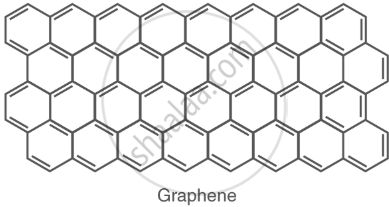 |
|
Fullerenes (C₆₀): spherical molecules used in nanotechnology. |
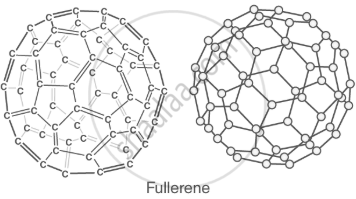 |
|
Carbon nanotubes: cylindrical structures with high strength and conductivity. |
 |
Bonding in Carbon Compounds
Ionic compounds have high melting and boiling points and conduct electricity in molten and dissolved states due to ionic bonds. In contrast, carbon compounds generally have lower melting and boiling points, as shown in the table below, indicating weaker intermolecular forces.
| Compound | Melting Point (°C) | Boiling Point (°C) |
|---|---|---|
| Methane (CH₄) | -183 | -162 |
| Ethanol (CH₃CH₂OH) | -117 | 78 |
| Chloroform (CHCl₃) | -64 | 61 |
| Acetic acid (CH₃COOH) | 17 | 118 |
Most carbon compounds do not conduct electricity, which suggests they lack ionic bonds and do not produce ions in solution.
Electronic Configuration of Carbon
- Atomic number: 6
- Electronic configuration: 2,4
- Valence electrons: 4
- Nearest noble gases: Helium (2), Neon (2,8)
Since carbon has four valence electrons, it can bond in three possible ways to achieve stability:
1. Losing four electrons to form C⁴⁺
- This would require excessive energy, making it unstable.
2. Gaining four electrons to form C⁴⁻
- The carbon nucleus would struggle to hold ten electrons, making this unstable.
3. Sharing four electrons with other atoms (Preferred method)
- Carbon forms covalent bonds, where atoms share electrons instead of transferring them.
Covalent Bonding and Electron-Dot Structures
A covalent bond forms when atoms share electrons to complete their valence shells, making them stable without forming ions. The electron-dot structure represents these shared electrons.
Examples of Covalent Bonding:
1. Hydrogen (H₂)
- Each hydrogen atom has one electron and needs one more to be stable.
- Two hydrogen atoms share electrons, forming a single bond (H—H).

Electron dot structure and line structure of hydrogen molecule with a single bond
2. Oxygen (O₂)
- Each oxygen atom has six valence electrons and needs two more.
- Two oxygen atoms share two pairs of electrons, forming a double bond (O=O).
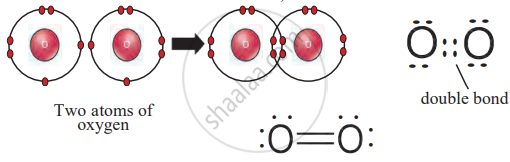
Double Bond
3. Nitrogen (N₂)
- Each nitrogen atom has five valence electrons and needs three more.
- Two nitrogen atoms share three pairs of electrons, forming a triple bond (N≡N).

Triple Bond
4. Methane (CH₄)
- Carbon needs four electrons to be stable.
- It shares one electron each with four hydrogen atoms, forming four single covalent bonds.
- Thus, covalent bonding allows carbon to form a wide variety of stable compounds.
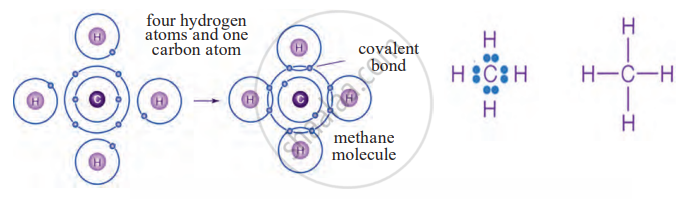
Electron-dot structure and line structure of methane molecule
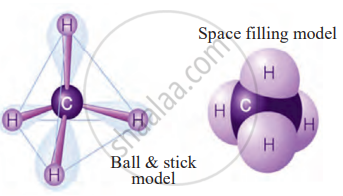
Models of methane molecule
Carbon Compounds and Their Uses
Types of Carbon-Based Compounds
- Hydrocarbons: carbon and hydrogen compounds (e.g., methane CH₄, ethane C₂H₆).
- Carbon-Oxygen Compounds: Includes CO₂, CO, and CO₃.
- Carbon-Sulphur Compounds: Examples include CS₂ and OCS.
- Carbon-Nitrogen Compounds: Includes Cyanogen (CN)₂ and Hydrogen Cyanide (HCN).
- Carbon-Halide Compounds: Examples: carbon tetrachloride (CCl₄), carbon tetrafluoride (CF₄).
Uses of Carbon and Its Compounds
- Life’s building block: essential for all living organisms.
- Fuels: Hydrocarbons are the primary energy source.
- Polymers: Used in plastics like polyethylene and polypropylene.
- Dyes and drugs: Carbon compounds are used in pharmaceuticals.
- Diamond: Used in jewellery and industrial cutting tools.
- Graphite: Used in pencils, lubricants, and electrodes.
- Glucose: A vital energy source for living cells.
Definition: Organic Compounds
All the compounds having carbon as a constituent element are called as organic compounds.
Definition: Covalent Bond
The chemical bond formed by sharing of two valence electrons between the two atoms is called covalent bond.
Key Points: Bonds in Carbon Compounds
- Organic compounds are mainly carbon compounds, except CO₂, CO, carbonates, bicarbonates, and carbides, which are inorganic.
- Carbon compounds have low melting and boiling points (generally below 300 °C) due to weak intermolecular forces.
- Most carbon compounds do not conduct electricity, showing the absence of ionic bonds.
- Carbon forms covalent bonds by sharing electrons to achieve a stable noble gas configuration.
- Carbon can form single, double, and triple covalent bonds, allowing the formation of many compounds like CH₄, O₂, and N₂.
