- Gravitational separation separates heavy ore particles from lighter gangue using water, based on difference in density (e.g., Wilfley table, hydraulic method).
- Magnetic separation uses an electromagnet to separate magnetic and non‑magnetic components of an ore (e.g., cassiterite).
- The froth flotation method is used for sulphide ores, where ore particles float on the froth due to their hydrophobic nature, while the gangue sinks.
- Vegetable oils (pine oil and eucalyptus oil) and air bubbles help form a froth that carries sulphide ore particles to the surface.
- Leaching involves dissolving the ore in a chemical solution (e.g., bauxite in NaOH), leaving gangue undissolved for separation.
Topics
Gravitation
- Concept of Gravitation
- Force and Motion
- Centripetal Force
- Kepler’s Laws
- Law of Orbit or Kepler's First Law
- Law of Areas or Kepler's Second Law
- Law of Periods or Kepler's Third Law
- Newton's Universal Law of Gravitation
- Uniform Circular Motion (UCM)
- Earth’s Gravitational Force
- Earth’s Gravitational Acceleration
- Mass and Weight
- Gravitational Waves
- Free Fall
- Gravitational Potential Energy
- Escape Velocity
- Weightlessness in Space
Periodic Classification of Elements
- Classification of Elements
- Dobereiner’s Triads
- Newland's Law of Octaves
- Mendeleev’s Periodic Table
- Insights into Mendeleev’s Periodic Table
- Modern Periodic Law
- The Modern Periodic Table
- Structure of the Modern Periodic Table
- Modern Periodic Table and Electronic Configuration of Elements
- Groups and Electronic Configuration
- Periods and Electronic Configuration
- Periodic Trends in the Modern Periodic Table
- Atomic Size
- Metallic and Non-metallic Characters
- Gradation in Halogen Family
Chemical Reactions and Equations
- Chemical Reaction
- Chemical Equations
- Balancing Chemical Equation
- Types of Chemical Reactions > Combination Reaction
- Types of Chemical Reactions > Decomposition Reaction
- Types of Chemical Reactions > Single Displacement Reaction
- Types of Chemical Reactions > Double Displacement Reaction
- Endothermic and Exothermic Processes
- Rate of Chemical Reaction
- Factors Affecting the Rate of a Chemical Reaction
- Chemical Properties of Carbon Compounds > Oxidation
- Chemical Properties of Carbon Compounds > Reduction
- Corrosion of Metals
- Rancidity
Effects of Electric Current
- Electric Circuit
- Heating Effect of Electric Current
- Magnetic Effect of Electric Current
- Right-hand Thumb Rule
- Applications of Biot-Savart's Law > Magnetic Field at the Centre of a Circular Loop
- Applications of Ampere’s Circuital Law > Magnetic Field of a Long Straight Solenoid
- Force on a Current Carrying Conductor in a Magnetic Field
- Fleming’s Left Hand Rule
- Electric Motor
- Electromagnetic Induction
- Galvanometer
- Faraday's Laws of Electromagnetic Induction
- Fleming’s Right Hand Rule
- Alternating current (AC) and Direct Current (DC)
- Electric Generator
Heat
Refraction of Light
Lenses
- Concept of Lenses
- Images Formed by Convex Lenses
- Images Formed by Concave Lenses
- Sign Convention
- Lens Formula
- Magnification
- Power of a Lens
- Combination of Lenses
- Defects of Vision and Their Corrections > Myopia
- Defects of Vision and Their Corrections > Hypermetropia
- Defects of Vision and Their Corrections > Presbyopia
- Apparent Size of an Object
- Use of Concave Lenses
- Use of Convex Lenses
- Persistence of Vision
Metallurgy
- Physical Properties of Metals
- Physical Properties of Non-metal
- Chemical Properties of Metal
- Reactions of Metals
- Reactivity Series of Metals
- Chemical Properties of Non-metal
- Ionic Compounds
- Metallurgy
- Basic Principles of Metallurgy > Concentration of Ores
- Basic Principles of Metallurgy > Extraction of Metals
- Basic Principles of Metallurgy > Refining of Metals
- Corrosion of Metals
- Prevention of Corrosion
Carbon Compounds
- Bonds in Carbon Compounds
- Carbon: A Versatile Element
- Hydrocarbons
- Straight chains, Branched chains, and Rings of Carbon atoms
- Functional Groups in Carbon Compounds
- Homologous Series
- Nomenclature
- Chemical Properties of Carbon Compounds > Combustion
- Chemical Properties of Carbon Compounds > Oxidation
- Chemical Properties of Carbon Compounds > Addition Reaction
- Chemical Properties of Carbon Compounds > Substitution Reaction
- Ethanol
- Ethanoic Acid
- Macromolecules and Polymers
Space Missions
School of Elements
The Magic of Chemical Reactions
- Chemical Equations
- Types of Chemical Reactions > Combination Reaction
- Types of Chemical Reactions > Decomposition Reaction
- Types of Chemical Reactions > Single Displacement Reaction
- Types of Chemical Reactions > Double Displacement Reaction
- Chemical Properties of Carbon Compounds > Oxidation
- Types of Double Displacement: Neutralization Reaction
The Acid Base Chemistry
- Properties of Acids > Physical Properties
- The pH Scale
- Acids, Bases and Their Reactivity
- Acid or a Base in a Water Solution
- Preparation and Uses of Baking Soda
- Preparation and Uses of Bleaching Powder
- Preparation and Uses of Washing Soda
- Preparation and Uses of Plaster of Paris
- Chemicals from Common Salt - Soap as a Salt
The Electric Spark
All about Electromagnetism
- Magnetic force
- Bar Magnet and Solenoid Analogy
- Right-hand Thumb Rule
- Applications of Biot-Savart's Law > Magnetic Field at the Centre of a Circular Loop
- Applications of Ampere’s Circuital Law > Magnetic Field of a Long Straight Solenoid
- Force on a Current Carrying Conductor in a Magnetic Field
- Electric Motor
- Electromagnetic Induction
- A.C. Generator
- Simple D.C. Motor
- Household Electrical Circuits
Wonders of Light 1
Wonders of Light 2
Striving for better Environment 1
- Abatement of Pollution
- Sustainable Use of Resources
- Definition: Concentration of Ores
- Key Points: Concentration of Ores
Maharashtra State Board: Class 10
Basic Principles of Metallurgy
Metallurgy involves the extraction of pure metals from ores through various processes. The first stage in this process is the concentration of ores, where impurities (gangue) are separated from the desired metal. The method used depends on the physical properties of the metal and gangue, the metal's reactivity, and environmental considerations.
1. Concentration of Ores: The removal of gangue from ores to increase the concentration of the desired metal. Different methods are used based on the properties of the ore and gangue.
a) Separation Based on Gravitation: Heavier ore particles can be separated from lighter gangue using gravity-based methods.
|
Wilfley Table Method:
|
 |
|
Hydraulic Separation Method:
|
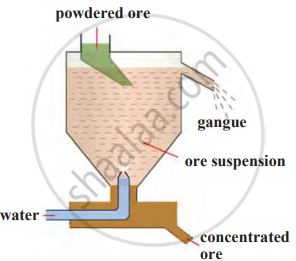 |
b) Magnetic Separation Method
- It is used when one component is magnetic and the other is not.
- The powdered ore is passed over a conveyor belt with two rollers:
- One is nonmagnetic, and the other is an electromagnetic.
- Magnetic metal particles stick to the magnetic roller while nonmagnetic gangue falls off.
- Example: Cassiterite (SnO₂) contains ferrous tungstate (FeWO₄), which is magnetic and can be separated.
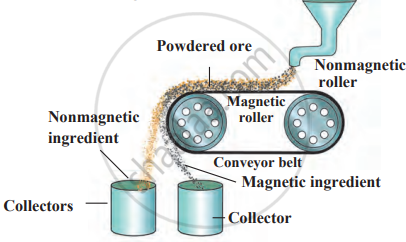
Magnetic separation
c) Froth Flotation Method
Based on the hydrophilic (water-attracting) and hydrophobic (water-repelling) properties of particles. Metal sulphides (ZnS, CuFeS₂) are hydrophobic and get coated with oil, while gangue is hydrophilic and remains in water.
Process:
- Finely ground ore is mixed with water and oil (e.g., pine or eucalyptus oil) in a tank.
- Air is blown, forming foam (froth).
- Sulphide ore particles attach to the froth and float, while gangue sinks.
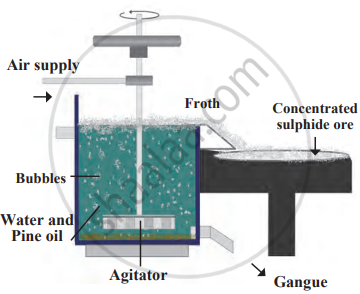
Froth floatation method
d) Leaching (Chemical Separation)
The ore is soaked in a specific solution that dissolves the metal but leaves gangue behind. Example: Bauxite (Al₂O₃) is purified using NaOH, which dissolves alumina but not impurities.
It is used for aluminium, gold, and silver extraction.
Maharashtra State Board: Class 10
Definition: Concentration of ores
The process of separating gangue from the ores is called concentration of ores.
Maharashtra State Board: Class 10
