Topics
Chemical Reactions and Equations
- Chemical Reactions in Daily Life
- Chemical Equations
- Balancing Chemical Equation
- Types of Chemical Reactions > Combination Reaction
- Types of Chemical Reactions > Decomposition Reaction
- Types of Chemical Reactions > Single Displacement Reaction
- Types of Chemical Reactions > Double Displacement Reaction
- Chemical Properties of Carbon Compounds > Oxidation
- The Effects of Oxidation Reactions in Everyday Life
Acids, Bases and Salts
- Acids and Bases in Daily Life
- Acids and Bases in the Laboratory
- Acids and Bases React with Metals
- Reaction of Metal Carbonates with Acids
- Acids and Bases Reaction with each other
- Reaction of Metallic Oxides with Acids
- Reaction of a Non-metallic Oxide with Base
- Common Properties of Acids and Bases
- The pH Scale
- Importance of pH in Everyday Life
- Salts > Family of Salts
- Salts > pH of Salts
- Salts > Chemicals from Common Salt
- Salts > Water in Salt Crystals
Metals and Non-metals
Carbon and its Compounds
- Importance of Carbon
- The Covalent Bond
- Allotropes of Carbon > Diamond
- Allotropes of Carbon > Graphite
- Allotropes of Carbon > Fullerene
- Carbon: A Versatile Element
- Organic Compounds
- Classification of Hydrocarbons
- Carbon Compounds: Chains, Branches, Rings
- Homologous Series
- Nomenclature
- Chemical Properties of Carbon Compounds > Combustion
- Ethanol
- Ethanoic Acid
- Soaps and Detergents
Life Processes
- Life Processes in Living Organisms
- Nutrition
- Autotrophic Nutrition
- Heterotrophic Nutrition
- Nutrition in Human Beings
- Dental Caries
- Production of ATP
- Blood Circulatory System
- Blood Vessels Entering and Leaving The Heart
- Valves of the Heart
- Tissue Fluid (Or Intercellular Fluid)
- Transportation in Plants
- Transportation of Water
- Excretion
- Excretion in Human Beings
- Kidney and Its Internal Structure
- Structure of a Kidney Tubule (Nephrons)
- Dialysis and Artificial Kidney
- Excretion in Plants
- Organ and Body Donation
Control and Co-ordination
How do Organisms Reproduce?
Heredity
Light – Reflection and Refraction
- Light and Its Straight-Line Propagation
- Reflection of Light
- Spherical Mirrors
- Image Formation by Spherical Mirrors
- Representation of Images Formed by Spherical Mirrors
- Image Formation by Concave Mirror
- Image Formation by a Convex Mirror
- Sign Convention for Reflection by Spherical Mirrors
- Ray Optics - Mirror Formula
- Refraction of Light
- Refraction through a Rectangular Glass Slab
- The Refractive Index
- Refraction by Spherical Lenses
- Image Formation by Lenses
- Image Formation in Lenses Using Ray Diagrams
- Sign Convention for Spherical Lenses
- Lens Formula
- Power of a Lens
The Human Eye and the Colourful World
Electricity
Magnetic Effects of Electric Current
- Magnetic Effect of Electric Current
- Applications of Biot-Savart's Law > Magnetic Field due to a Finite Straight Current-Carrying Wire
- Magnetic Field Due to a Current-Carrying Conductor
- Right-hand Thumb Rule
- Applications of Biot-Savart's Law > Magnetic Field at the Centre of a Circular Loop
- Applications of Ampere’s Circuital Law > Magnetic Field of a Long Straight Solenoid
- Force on a Current Carrying Conductor in a Magnetic Field
- Fleming’s Left Hand Rule
- Magnetism in Medicine
- Domestic Electric Circuits
Our Environment
- Definition: Electropositive Elements
- Key Points: Chemical Properties of Metal
Chemical Properties
1. Reactivity of Metals: Metals are very reactive and easily combine with other substances like oxygen, water, or acids.
2. Losing Electrons: When metals react, they lose electrons (tiny negatively charged particles in an atom). By losing electrons, the metal becomes positively charged.
3. Positively charged ions: Metals lose electrons and turn into positively charged ions. For example, when sodium reacts with water, it loses electrons and becomes a positively charged sodium ion.
4. Electropositive Elements: Metals are called electropositive elements because they easily lose electrons and become positively charged. This means metals have a tendency to give away their electrons in a chemical reaction.
5. Electronic Configuration: Electronic configuration is the basis of the chemical behaviour of elements. Most metals have up to three electrons in their outermost shell.
| Metal | Atomic number | Electronic configuration |
|---|---|---|
| ₁₁Na | 11 | 2, 8, 1 |
| ₁₂Mg | 12 | 2, 8, 2 |
| ₁₃Al | 13 | 2, 8, 3 |
6. Formation of Ions: Metals have a tendency to lose their valence electrons to form positively charged ions, called cations.
Na → Na⁺ + e⁻
(2, 8, 1) (2, 8)
Sodium → Sodium ion
Mg → Mg²⁺ + 2e⁻
(2, 8, 2) (2, 8)
Magnesium → Magnesium ion
Al → Al³⁺ + 3e⁻
(2, 8, 3) (2, 8)
Aluminium → Aluminium ion
7. Reaction with Oxygen: Metals combine with oxygen to form their oxides.
Metal + Oxygen → Metal oxide
The metal oxides are basic in nature. Metal oxides react with acids to form salt and water.
Metal oxide + Acid → Salt + Water
8. Reaction with Acid: Take dilute hydrochloric acid in a test tube. Add zinc dust to it. Take a glowing splinter near the mouth of the tube and observe. Most metals react with dilute acids to form metal salts, releasing hydrogen gas.
Metal + Dilute acid → Salt + Hydrogen gas.
9. Reaction with Water: Most metals do not react visibly and rapidly with cold water. However, some metals like sodium and potassium react with cold water to produce their hydroxides and hydrogen gas. Magnesium requires steam for a similar reaction.
Experiment
1. Aim: To observe the reaction of different metals when heated in a flame.
2. Requirements
- Apparatus: Pair of tongs or spatula, knife, burner
- Chemicals: Aluminium, copper, iron, lead, magnesium, zinc, and sodium (handle sodium carefully under supervision)
3. Procedure
- Hold each metal sample at the top of the flame using tongs or a spatula.
- Observe which metal catches fire readily.
- Note any changes in the metal’s surface and the flame’s colour while burning.
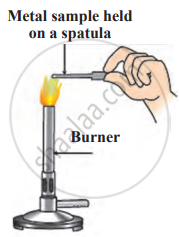
Combustion of metal
4. Conclusion: Some metals, like magnesium and sodium, catch fire quickly, while others do not burn easily. The surface of the burning metal may change, forming an oxide layer. Different metals produce different flame colours when burnt.
Definition: Electropositive Elements
Metals are reactive. They lose electrons easily and become positively charged ions. That is why metals are called electropositive elements.
Key Points: Chemical Properties of Metal
- Most metals form basic oxides with oxygen; aluminium and zinc oxides are amphoteric.
- Reactivity with oxygen varies; sodium and potassium react quickly and are kept in kerosene.
- Metals may react with water to form hydroxides and release hydrogen gas, depending on temperature.
- Metals react with dilute acids to give salt and hydrogen gas; copper and nitric acid are exceptions.
- More reactive metals can displace less reactive ones; this forms the basis of the reactivity series.
