Topics
Chemical Reactions and Equations
- Chemical Reactions in Daily Life
- Chemical Equations
- Balancing Chemical Equation
- Types of Chemical Reactions > Combination Reaction
- Types of Chemical Reactions > Decomposition Reaction
- Types of Chemical Reactions > Single Displacement Reaction
- Types of Chemical Reactions > Double Displacement Reaction
- Chemical Properties of Carbon Compounds > Oxidation
- The Effects of Oxidation Reactions in Everyday Life
Acids, Bases and Salts
- Acids and Bases in Daily Life
- Acids and Bases in the Laboratory
- Acids and Bases React with Metals
- Reaction of Metal Carbonates with Acids
- Acids and Bases Reaction with each other
- Reaction of Metallic Oxides with Acids
- Reaction of a Non-metallic Oxide with Base
- Common Properties of Acids and Bases
- The pH Scale
- Importance of pH in Everyday Life
- Salts > Family of Salts
- Salts > pH of Salts
- Salts > Chemicals from Common Salt
- Salts > Water in Salt Crystals
Metals and Non-metals
Carbon and its Compounds
- Importance of Carbon
- The Covalent Bond
- Allotropes of Carbon > Diamond
- Allotropes of Carbon > Graphite
- Allotropes of Carbon > Fullerene
- Carbon: A Versatile Element
- Organic Compounds
- Classification of Hydrocarbons
- Carbon Compounds: Chains, Branches, Rings
- Homologous Series
- Nomenclature
- Chemical Properties of Carbon Compounds > Combustion
- Ethanol
- Ethanoic Acid
- Soaps and Detergents
Life Processes
- Life Processes in Living Organisms
- Nutrition
- Autotrophic Nutrition
- Heterotrophic Nutrition
- Nutrition in Human Beings
- Dental Caries
- Production of ATP
- Blood Circulatory System
- Blood Vessels Entering and Leaving The Heart
- Valves of the Heart
- Tissue Fluid (Or Intercellular Fluid)
- Transportation in Plants
- Transportation of Water
- Excretion
- Excretion in Human Beings
- Kidney and Its Internal Structure
- Structure of a Kidney Tubule (Nephrons)
- Dialysis and Artificial Kidney
- Excretion in Plants
- Organ and Body Donation
Control and Co-ordination
How do Organisms Reproduce?
Heredity
Light – Reflection and Refraction
- Light and Its Straight-Line Propagation
- Reflection of Light
- Spherical Mirrors
- Image Formation by Spherical Mirrors
- Representation of Images Formed by Spherical Mirrors
- Image Formation by Concave Mirror
- Image Formation by a Convex Mirror
- Sign Convention for Reflection by Spherical Mirrors
- Ray Optics - Mirror Formula
- Refraction of Light
- Refraction through a Rectangular Glass Slab
- The Refractive Index
- Refraction by Spherical Lenses
- Image Formation by Lenses
- Image Formation in Lenses Using Ray Diagrams
- Sign Convention for Spherical Lenses
- Lens Formula
- Power of a Lens
The Human Eye and the Colourful World
Electricity
Magnetic Effects of Electric Current
- Magnetic Effect of Electric Current
- Applications of Biot-Savart's Law > Magnetic Field due to a Finite Straight Current-Carrying Wire
- Magnetic Field Due to a Current-Carrying Conductor
- Right-hand Thumb Rule
- Applications of Biot-Savart's Law > Magnetic Field at the Centre of a Circular Loop
- Applications of Ampere’s Circuital Law > Magnetic Field of a Long Straight Solenoid
- Force on a Current Carrying Conductor in a Magnetic Field
- Fleming’s Left Hand Rule
- Magnetism in Medicine
- Domestic Electric Circuits
Our Environment
- Definition: Covalent Compound
- Definition: Covalent Molecule
- Key Points: The Covalent Bond
Introduction of Covalent Bond:
A covalent bond is a chemical bond formed when two atoms share their valence electrons to complete their electron octet or duplet. This sharing of electrons allows both atoms to achieve stability.
- Covalent bonds are typically formed between atoms with similar electronegativities, as they do not tend to transfer electrons.
- Unlike ionic bonds, where electrons are transferred, covalent bonds involve the sharing of valence electrons.
- The shared electrons count toward the outer shell of both atoms, allowing them to fulfil the octet rule (for most atoms) or the duplet rule (for smaller atoms like hydrogen).
Formation of Covalent Bonds
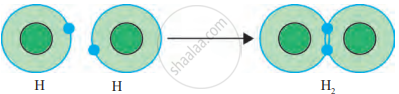
(a) Formation of H₂ Molecule (Hydrogen Molecule):
- Each hydrogen atom has 1 electron in its valence shell.
- Hydrogen needs 1 more electron to complete its duplet.
- Two hydrogen atoms share their single electrons, forming a single covalent bond. The duplet of both hydrogen atoms is complete.
- The molecule is represented as H—H, where the dash represents the covalent bond.
H • + H • → H:H → H—H
Formation of Covalent bond in the H₂ molecule
(b) Formation of H₂O Molecule (Water):
- Oxygen has 6 valence electrons in its outer shell.
- It needs 2 more electrons to complete its octet.
- Each hydrogen atom has 1 valence electron and needs 1 more electron to complete its duplet.
- The oxygen atom shares 1 electron each with 2 hydrogen atoms, forming two covalent bonds. The octet of the oxygen atom is completed.
- The duplets of the two hydrogen atoms are also completed.
The molecule is represented as H—O—H. 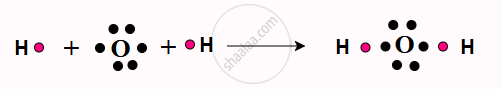

Formation of Covalent bond in H₂O molecule
(c) Formation of N₂ Molecule (Nitrogen Molecule):
- Each nitrogen atom has 5 valence electrons in its outer shell.
- Nitrogen needs 3 more electrons to complete its octet.
- Two nitrogen atoms share three pairs of electrons (6 electrons in total) to complete their octets.
- This forms a triple covalent bond between the two nitrogen atoms. Both nitrogen atoms achieve a stable octet.
- The molecule is represented as N≡N, where the three dashes represent the triple bond.
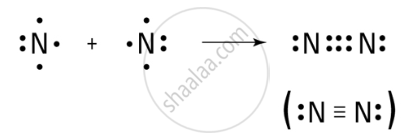
Formation of Covalent bond in the N₂ molecule
Properties of Covalent Compounds:
- Covalent compounds are generally poor conductors of electricity, as they do not have free ions or electrons.
- They have low melting and boiling points due to weak intermolecular forces, despite strong covalent bonds.
- Most covalent compounds are insoluble in water but dissolve in organic solvents like alcohol and benzene.
- They can exist in all three states of matter: solid, liquid, or gas, depending on their molecular structure.
- Covalent compounds are typically brittle and not malleable or ductile.
- They have definite molecular structures with fixed ratios of atoms.
- Reactions involving covalent compounds are often slower compared to ionic compounds due to the breaking and rearranging of covalent bonds.
- They may exhibit polar or non-polar properties, depending on the electronegativity difference between the bonded atoms.
Key Points: The Covalent Bond
- Carbon forms covalent bonds by sharing electrons to achieve a noble gas configuration.
- Covalent bonds can be single, double, or triple, as seen in molecules like H₂, O₂, and N₂.
- Covalent compounds have low melting and boiling points and are poor conductors of electricity.
- Carbon has allotropes such as diamond, graphite, and fullerene (C₆₀), each with different physical properties.
Definition: Covalent Compound
The chemical bond that is formed between two combining atoms by mutual sharing of one or more pairs of electrons is called a covalent (or a molecular) bond, and the compound formed due to this bond is called a covalent compound.
Definition: Covalent Molecule
The molecule formed due to the sharing of electrons (covalent bond) is called a covalent molecule.