Advertisements
Advertisements
प्रश्न
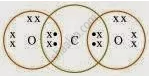
What would be the electron dot structure of carbon dioxide which has the formula CO2?
Advertisements
उत्तर
APPEARS IN
संबंधित प्रश्न
Butanone is a four-carbon compound with the functional group:
Give appropriate scientific reasons for Carbon tetrachloride does not conduct electricity.
Fill in the blank in the following sentence:
The number of single covalent bonds in C2H2 molecule are ...........
Why does carbon form compounds mainly by covalent bonding?
Draw the electron-dot structure of HCl compound and state the type of bonding.
Write two points of difference in the structures of diamond and graphite.
Describe the structure of graphite with the help of a labelled diagram.
The solution of one of the following compounds will not conduct electricity. This compounds is:
(a) NaCl
(b) CCl4
(c) MgCl2
(d) CaCl2
Methane molecule is non-polar molecule. Explain.
Write answer as directed.
What causes the existance of very large number of carbon compound ?
Give examples for the following:
Two liquid non polar compounds.
Explain the structure of Hydronium ion.
Explain why Carbon tetrachloride does not dissolve in water.
The following table shows the electronic configuration of the elements W, X, Y, Z:
|
Element |
W |
X |
Y |
Z |
|
Electronic |
2,8,1 |
2,8,7 |
2,5 |
1 |
Answer the following questions based on the table above:
What type of bond is formed between Y and Z.
Electrons are getting added to an element Y:
Is Y getting oxidised or reduced?
What do you understand by dipole (polar) molecule?
Write an Explanation.
Alkane
Consider the coordination compound, K2[Cu(CN)4]. A coordinate covalent bond exists between:
The correct structural formula of butanoic acid is -
Carbon can neither form C4- cation nor C4 anion. Why?
