Advertisements
Advertisements
प्रश्न
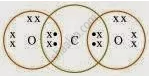
What would be the electron dot structure of carbon dioxide which has the formula CO2?
Advertisements
उत्तर
APPEARS IN
संबंधित प्रश्न
What would be the electron dot structure of a molecule of sulphur which is made up of eight atoms of sulphur? (Hint – the eight atoms of sulphur are joined together in the form of a ring.)
Ethane, with the molecular formula C2H6 has ______.
State whether the following statement is true or false:
Diamond and graphite are the covalent compounds of carbon element (C)
What do you call the compounds having the same molecular formula but different structural arrangements of atoms?
What type of bonds are present in H2 molecule? Draw their electron-dot structures.
Fill in the following blank with suitable word:
The form of carbon which is known as black lead is ...........
Draw the electron-dot structure of HCl compound and state the type of bonding.
A covalent molecule having a double bond between its atoms is:
(a) Hydrogen
(b) Oxygen
(c) water
(d) ammonia
The pencil leads are made of mainly:
(a) lithium
(b) charcoal
(c) lead
(d) graphite
The number of isomers formed by the hydrocarbon with molecular formula C5H12 is:
(a) 2
(b) 5
(c) 3
(d) 4
Give the characteristic properties of covalent compounds.
Which element exhibits the property of catenation to maximum extent and why?
Choose and write the correct option.
Which type of carbon-carbon bonds are present in Vanaspati ghee?
Explain the following briefly:
Cl2 is a non polar molecule, while HCl is a polar molecule.
Explain why carbon tetrachloride does not dissolve in water.
Draw the electron dot structure of covalent compound methane (non polar) and HCL (polar) and give two difference between them.
What is the difference between a polar covalent compound and a non-polar covalent compound?
Complete the following activity.
Write the names of the hydrocarbons for the following structural formula.
(isobutylene, cyclohexane, propene, cyclohexene, cyclopentane, benzene, propyne, isobutane, propane)
| \[\begin{array}{cc}\phantom{......}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{..}\\ \phantom{.....}|\phantom{....}|\phantom{....}|\\ \ce{H - C - C = C}\\\phantom{.....}|\phantom{.........}|\\ \phantom{.....}\ce{H}\phantom{........}\ce{H}\end{array}\] |
Consider the coordination compound, K2[Cu(CN)4]. A coordinate covalent bond exists between:
