Advertisements
Advertisements
Question
What would be the electron dot structure of carbon dioxide which has the formula CO2?
Advertisements
Solution
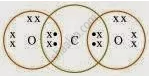
APPEARS IN
RELATED QUESTIONS
Ethane, with the molecular formula C2H6 has ______.
Give appropriate scientific reasons for Carbon tetrachloride does not conduct electricity.
What type of bonds are present in Cl2 molecule? Draw their electron-dot structures.
Give one example of a molecule containing a single covalent bond.
Fill in the following blank with suitable word:
The general formula CnH2n for cycloalkanes is the same as that of ...........
Explain why covalent compounds have generally high melting points?
What is buckminsterfullerene?
Why is graphite used for making dry cell electrodes but diamond is not?
State any two uses of diamond.
What is graphite?
State the type of bonding in the following molecule.
Methane
Give two example in following case:
Gaseous polar compounds
Choose the correct answer from the options given below
Condition favorable for formation of a covalent bond is
(a) Compound X consists of molecules.
Choose the letter corresponding to the correct answer from the choices (a), (b), (c) and (d) given below
The type of bonding in X will be
Compound X consists of molecules.
Choose the letter corresponding to the correct answer from the options A, B, C and D given below:
The type of bonding in X will be ______.
Acids dissolve in water and produce positively charged ions. Draw the structure of these positive ions.
State the type of bond formed when the combining atom has zero E.N. difference.
Fill in the blank and rewrite the completed statement:
Covalent compounds are generally soluble in _________ solvents.
Your body is made up of carbon.
Complete the following activity.
Write the names of the hydrocarbons for the following structural formula.
(isobutylene, cyclohexane, propene, cyclohexene, cyclopentane, benzene, propyne, isobutane, propane)
 |
