Advertisements
Advertisements
प्रश्न
Explain Hoffmann’s exhaustive alkylation with suitable reactions.
Advertisements
उत्तर
Hofmann’s exhaustive alkylation of amines:
- When a primary amine is heated with excess of primary alkyl halide it gives a mixture of secondary amine, tertiary amine along with tetraalkylammonium halide. This can be given as,
\[\ce{\underset{\text{1° Amine}}{R - NH2} ->[R - X][-HX] \underset{\text{2° Amine}}{R2NH} ->[R - X][-HX] \underset{\text{3° Amine}}{R3N} ->[R - X][-HX] \underset{\text{Tetraalkyl ammonium halide}}{R4N+X-}}\] - If the excess alkyl halide is used tetraalkylammonium halide is obtained as a major product and the reaction is known as exhaustive alkylation of amines.
- Tetraalkylammonium halides or quaternary ammonium salts are the derivatives of ammonium salts in which all the four hydrogen atoms attached to nitrogen in N+H4 are replaced by four alkyl groups (same or different).
- Tetraalkylammonium halides are crystalline solids.
- Primary, secondary and tertiary amines consume three, two and one moles of alkyl halide respectively to get converted into quaternary ammonium salt.
- The reaction is carried out in presence of mild base NaHCO3, to neutralize the large quantity of HX formed.
- If the alkyl halide is methyl iodide, the reaction is called exhaustive methylation of amines.
e.g. When methylamine is heated with excess methyl iodide, it gives tetramethyl ammonium iodide.
\[\ce{\underset{\text{Methylamine}}{CH3 - NH2} + \underset{\text{Methyl iodide}}{CH3 - I} ->[\Delta] \underset{\text{Dimethylamine}}{(CH3)2NH} + HI}\]
\[\ce{(CH3)2 - NH + CH3 - I ->[\Delta] \underset{\text{Trimethyl amine}}{(CH3)3N} + HI}\]
\[\ce{(CH3)3N + CH3 - I ->[\Delta] \underset{\text{Tetramethyl ammonium iodide}}{(CH3)4N+I-}}\]
संबंधित प्रश्न
Write a short note on Hoffmann bromamide degradation.
Illustrate the following reaction giving suitable example in each case:Gabriel phthalimide synthesis
How do you convert the following: C6H5CONH2 to C6H5NH2
Accomplish the following conversion:
Benzamide to toluene
Give the structure of A, B and C in the following reaction:
\[\ce{C6H5N2Cl ->[CuCN] A ->[H2O/H+] B ->[NH3][\Delta] C}\]
Give the structure of A, B and C in the following reaction:
\[\ce{C6H5NO2 ->[Fe/HCl] A ->[NaNO2 + HCl][273 K] B ->[H2O/H+][\Delta] C}\]
Why cannot aromatic primary amines be prepared by Gabriel phthalimide synthesis?
Write the reaction of aromatic primary amine with nitrous acid.
Write the reaction of aliphatic primary amine with nitrous acid.
Give a plausible explanation for the following:
Why are amines less acidic than alcohols of comparable molecular masses?
Account for the following:
Gabriel phthalimide synthesis is not preferred for preparing aromatic primary amines.
Choose the most correct option.
Which of the following compounds will dissolve in aqueous NaOH after undergoing reaction with Hinsberg reagent?
Write reactions to prepare ethanamine from Acetonitrile.
Write reactions to bring about the following conversions.
Acetamide to methylamine
Why cannot aniline be prepared by Gabriel phthalimide synthesis?
Acetamide on reduction using Na/C2H5OH gives ____________.
The end product C of the following reaction is
\[\ce{C2H5NH2 ->[HNO2] A ->[PCl5] B ->[NH3][Alcohol] C}\]
Identify the product obtained when benzyl chloride undergoes ammonolysis in presence of excess ammonia followed by the reaction with two moles of methyl iodide.
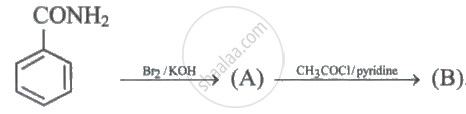
Identify the major product (B).
Which of the following reactions is appropriate for converting benzamide to aniline?
Identify the INCORRECT statement regarding Hofmann bromamide reaction.
In aqueous phase the order of basic strength of alkylamine is ______.
Which of the following amines cannot be prepared by Gabriel phthalimide synthesis?
Identify product B in the following reaction.
\[\ce{Aniline ->[NaNO2][HCl] A ->[KI] B}\]
Which of the following reagents is used in Hofmann's elimination reaction of amines?
Which of the following compounds is obtained when quaternary ammonium hydroxide is strongly heated?
Which of the following does NOT give carbylamine test?
Given below are two statements labelled as Assertion (A) and Reason (R).
Assertion (A): Alkyl halides are insoluble in water.
Reason (R): Alkyl halides have halogen attached to sp3 hybrid carbon.
Select the most appropriate answer from the options given below:
Amongst the following, the strongest base in aqueous medium is ______.
Which of the following reagents would not be a good choice for reducing an aryl nitro compound to an amine?
The source of nitrogen in Gabriel synthesis of amines is ______.
Amongst the given set of reactants, the most appropriate for preparing 2° amine is ______.
Reduction of aromatic nitro compounds using \[\ce{Fe}\] and \[\ce{HCl}\] gives ______.
Best method for preparing primary amines from alkyl halides without changing the number of carbon atoms in the chain is ______.
Which of the following compounds is the weakest Brönsted base?
Among the following amines, the strongest Brönsted base is:
Which of the following methods of preparation of amines will give same number of carbon atoms in the chain of amines as in the reactant?
Which of the following amines can be prepared by Gabriel synthesis.
(i) Isobutyl amine
(ii) 2-Phenylethylamine
(iii) N-methylbenzylamine
(iv) Aniline
Which of the following reactions are correct?
(i)

(ii)

(iii)

(iv)

Identify A and B in the following reaction.

How will you bring out the following conversion?

How will you carry out the following conversions?

Match the reactions given in Column I with the statements given in Column II.
| Column I | Column II | ||
| (i) | Ammonolysis | (a) | Amine with lesser number of carbon atoms |
| (ii) | Gabriel phthalimide synthesis | (b) | Detection test for primary amines. |
| (iii) | Hoffmann Bromamide reaction | (c) | Reaction of phthalimide with \[\ce{KOH}\] and \[\ce{R-X}\] |
| (iv) | Carbylamine reaction | (d) | Reaction of alkylhalides with \[\ce{NH3}\] |
Describe Gabriel's phthalimide synthesis. (Give reaction)
Account for the following:
Aniline cannot be prepared by the ammonolysis of chlorobenzene under normal conditions.
A primary amine is formed by an amide on treatment with bromine and alkali. The primary amine has
Which of the following reactions will not give a primary amine?
Which of the following compound gives pink colour on reaction with phthalic anhydride in cone. H2SO4 followed by treatment with NaOH?
Which of the following reaction DOES NOT involve Hoffmann bromamide degradation?
- Phenyl methenamine
- N, N - Dimethylaniline
- N - Methyl aniline
- Benzenamine
Choose the correct order of the basic nature of the above amines.
Which of the following would not be a good choice for reducing nitrobenzene to aniline?
